This is a SUPER easy guide on sodium element.
In fact, the table mentioned below is the perfect information box (Which gives you every single detail about the Sodium element in Periodic table.)
So if you want to know anything about Sodium element, then this guide is for you.
Let’s finish this very quickly.
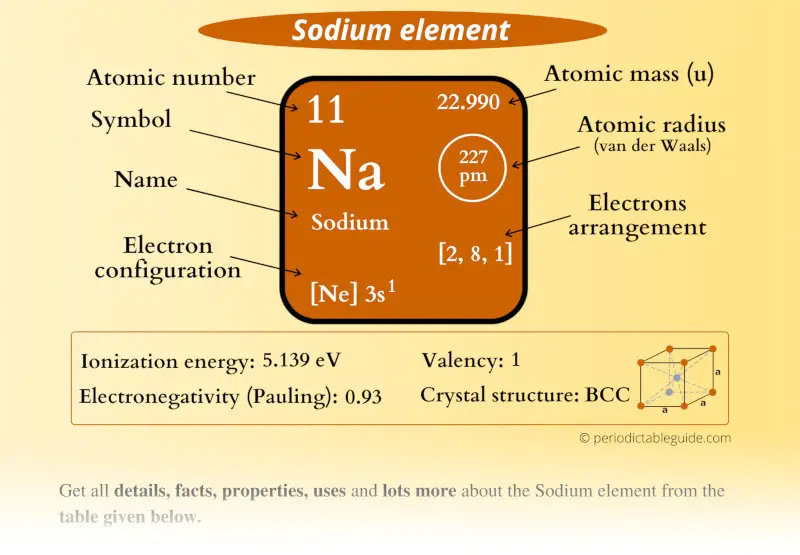
Sodium Element (Na) Information
| Appearance |  Silvery white |
| State (at STP) | Solid |
| Position in periodic table | 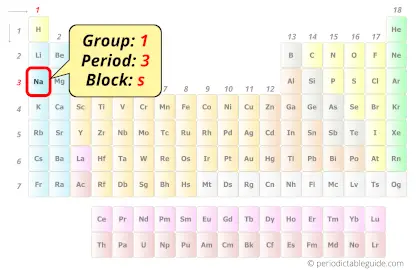 Group: 1, Period: 3, Block: s |
| Category | 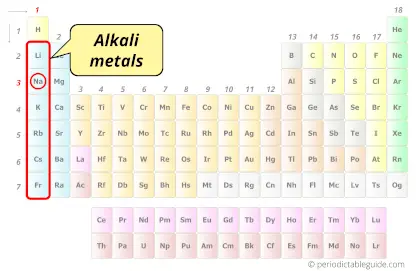 Alkali metals |
| Atomic number or Protons | 11 |
| Neutrons | 12 |
| Electrons | 11 |
| Symbol | Na |
| Atomic mass | 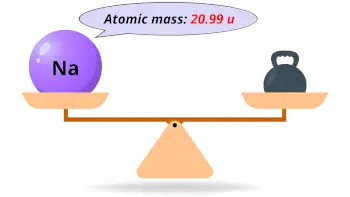 22.990 u |
| Electrons arrangement or Bohr model | 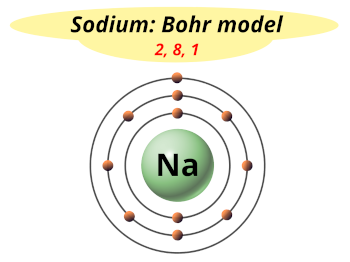 2, 8, 1 |
| Electronic configuration | [Ne] 3s1 |
| Atomic radius | 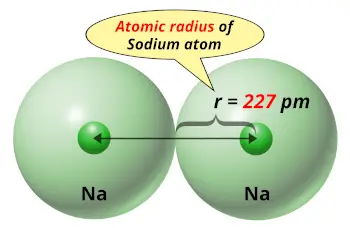 227 picometers (van der Waals radius) |
| Valence electrons | 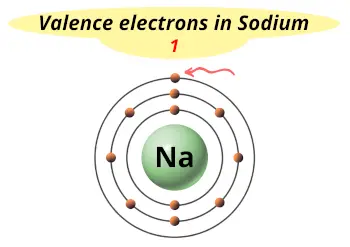 1 |
| 1st Ionization energy | 5.139 eV |
| Electronegativity | 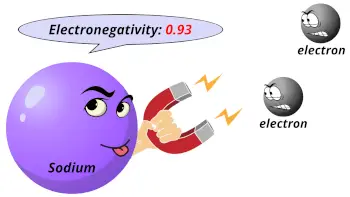 0.93 (Pauling scale) |
| Crystal structure | 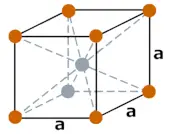 BCC (Body Centered Cubic) |
| Melting point | 370.9 K or 97.7 °C or 208 °F |
| Boiling point | 1156 K or 882.9 °C or 1621.2 °F |
| Density | 0.97 g/cm3 |
| Main isotope | 23Na |
| Who discovered Sodium and when? |  Sir Humphry Davy in 1807 |
| CAS number | 7440-23-5 |
Sodium in Periodic table
Sodium element is in group 1 and period 3 of the Periodic table. Sodium is the s-block element and it belongs to alkali metals group.
| H | He | ||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La* | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac** | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| *Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | ||||
| **Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
←Move to: Neon (Ne) element – Periodic Table
→Move to: Magnesium (Mg) element – Periodic Table
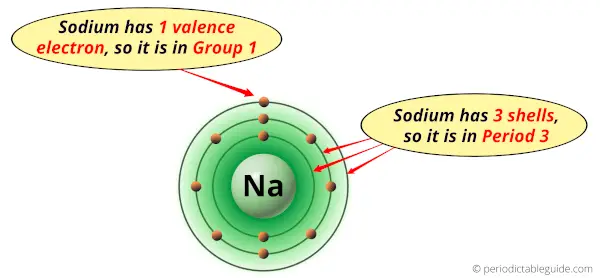
Why is Sodium in Group 1 and Period 3 of the Periodic table?
Sodium is in group 1 because it has 1 valence electron.
Sodium is in period 3 because it has 3 shells or orbits.
Now I’ll explain to you the perfect detailed reason why sodium is in group 1, why it is in period 3 and also why it is in s-block?
Let’s see the reasons one by one.
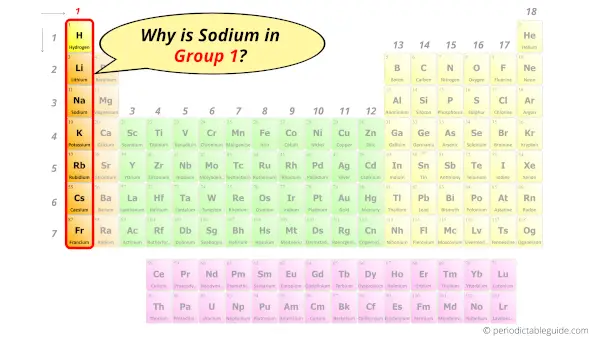
Why is Sodium in Group 1?
Do you know, how many electrons can be accommodated in the first shell, second shell, third shell, fourth shell, etc…?
Here is the table showing the capacity of orbits to hold electrons.
Number of electrons in shells.
| Orbit / Shell (n) | Maximum no. of electrons this orbit can hold |
| K shell, n = 1 | 2 × 1² = 2 |
| L shell, n = 2 | 2 × 2² = 8 |
| M shell, n = 3 | 2 × 3² = 18 |
| N shell, n = 4 | 2 × 4² = 32 |
Thus,
- 1st shell can hold 2 electrons.
- 2nd shell can hold 8 electrons.
- 3rd shell can hold 18 electrons.
- 4th shell can hold 32 electrons.
Now the atomic number of Sodium (Na) is 11.
Hence sodium element has the electrons arrangement 2, 8, 1.
This electron arrangement indicates that the outermost orbit of Sodium (Na) has 1 electron.
Hence, it lies in group 1.
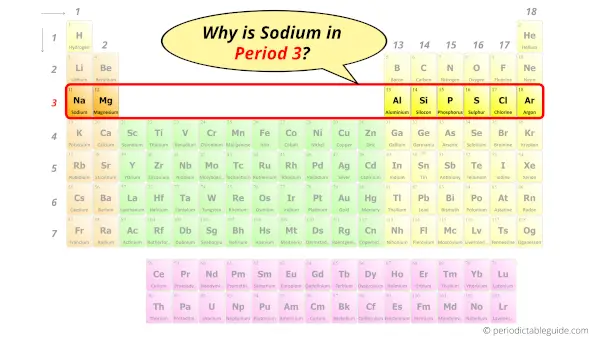
Why is Sodium in Period 3?
Let me ask you a question.
How many shells does sodium have?
It’s 3. Right?
You have already seen the bohr model of sodium element in the above table.
From the Bohr model, it can be found that the number of orbits or shells in sodium is 3.
Hence, as sodium has 3 orbits, it lies in period 3 of the Periodic table.
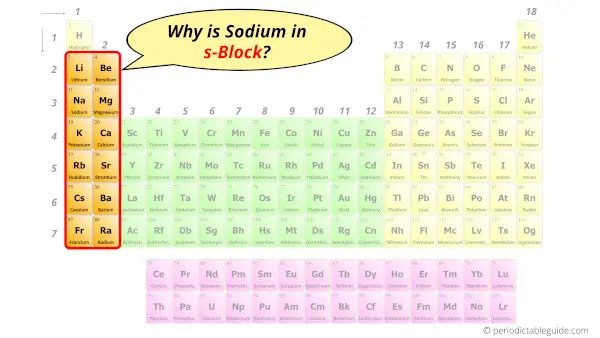
Why is Sodium in s-block?
Before knowing this reason, first of all a simple question to you.
How can you determine the blocks wise position of elements?
The simple answer: The elements will lie in the s, p, d or f block will completely depend upon the subshell in which the last electron will enter.
For example; the electron configuration of Sodium is [Ne] 3s1.
So the last electron of Sodium enters the s-subshell or s-orbital.
Hence, sodium is the s-block element.
Is Sodium a Metal or Nonmetal?
Yes, Sodium is a Metal. It’s a silvery white colored reactive metal. It is a soft metal and it can be cut even by using a kitchen knife. It is a light metal and it even floats on water.

But do you know what exactly the metals are?
- Metals are the elements which lose electron/s during a chemical reaction.
In other words, metals are electron donors.
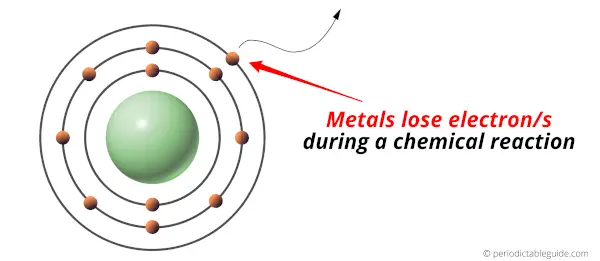
Metals are found on the left side of the Periodic table.
For more clarification, see the location of metals on the Periodic table (Image).
Why Sodium catches fire in water?

It seems very dangerous. Isn’t it?
Let me explain the reasons why sodium catches fire in water.
Or in other words, why is sodium a very reactive metal?
The reasons are;
- Large atomic size
- One valence electron
- Weak metallic bonds
Let me tell you few things about these three reasons (very quickly).
1). Large atomic size
Sodium element is in group 1, and group 1 elements have the larger atomic radius compared to elements of other groups.
Due to larger atomic radius, they have a weak attractive force for the outermost electron.
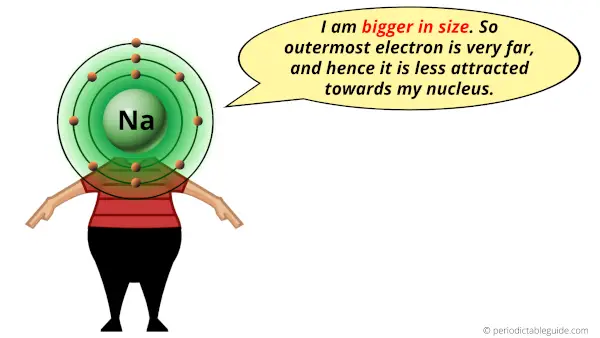
And due to weak attractive force, the outermost electron gets easily escaped during a chemical reaction.
Hence sodium metal reacts vigorously with the water and it even catches fire.
2). One valence electron
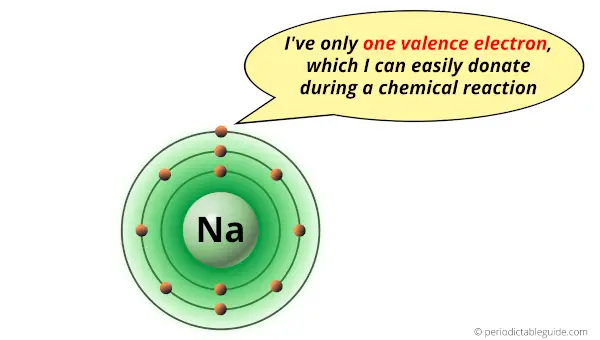
The sodium atom have only one electron in outermost orbit. This electron gets easily lost or shared during a chemical reaction.
Hence due to rapid losing of electrons, the sodium metal is highly reactive.
3). Weak metallic bonds
You know that sodium is a metal, but the fact is that it has weak metallic bonds.
In other words, the attractive force among the sodium atoms is weak.
In short sodium element has less intermolecular force of attraction.
Due to less intermolecular force of attraction, it loses electrons very easily and hence it reacts with water very fast.
Also see: Why sodium is called alkali metal?
I hope you have understood the reason why sodium is very reactive to water.
Now let’s move to another interesting question.
Why Sodium gives yellow flame on heating?

When sodium metal is heated with a flame, its outermost electron gets excited onto a higher energy level.
When this excited electron loses its energy, it again comes back to the lower energy level.
During this phenomenon, it emits an electromagnetic radiation having a wavelength of around 589 nm.
This emitted radiation of 589 nm is nothing but a visible golden yellow light.
Hence, sodium gives golden yellow flame when it is heated.
Why Sodium floats on water?

You might have studied the concept of density in physics.
By knowing the density of two objects, we can come to know which object will float and which will sink in water.
If the object has a density higher than the water, then it will sink. And if the density of object is less than the water, then it will float on water.
Now let’s see about sodium metal and it’s density.
Sodium has a density of 0.97 g/cm3, while water has a density of 1 g/cm3.
That means sodium metal is less dense than water.
Because of this reason, the sodium metal floats on water.
8 Interesting facts about Sodium
Here are the interesting facts about Sodium element.
- Sodium is a soft metal and it can be cut even by using a kitchen knife.
- Sodium metal is silver white in color when it is freshly cut, but it suddenly forms an oxide layer if kept open in air.
- Sodium metal is lighter than water, hence it floats on water.
- Sodium burns with a golden yellow flame when it is heated.
- Sodium element is also essential to maintain the fluid balance in the cells of our body.
- Around 2.6% of the Earth’s crust contains sodium metal. Out of all alkali metals, Sodium is most abundant.
- In nature, Sodium never exists as a pure metal because of its high reactivity. It is always found in a compound form with other elements.
Sodium has many isotopes, out of which only one isotope is most stable (i.e 23Na).
Explore our New Interactive Periodic Table (with Rotating Bohr Models and More)

Details about this Periodic table:
- Access detailed info on all elements: atomic mass, electron configurations, charges, and more.
- View rotating Bohr models for all 118 elements.
- Get a free HD image of the Periodic Table.
Note: For future use, bookmark this Periodic table or visit “PeriodicTableGuide.com”
External Resources:
- P. (n.d.). Sodium | Na (Element) – PubChem. Sodium | Na (Element) – PubChem. https://pubchem.ncbi.nlm.nih.gov/element/Sodium
- Sodium – Element information, properties and uses | Periodic Table. (n.d.). Sodium – Element Information, Properties and Uses | Periodic Table. https://www.rsc.org/periodic-table/element/11/sodium
- Sodium – Wikipedia. (2016, October 15). Sodium – Wikipedia. https://en.wikipedia.org/wiki/Sodium
- It’s Elemental – The Element Sodium. (n.d.). It’s Elemental – the Element Sodium. https://education.jlab.org/itselemental/ele011.html
