This is a SUPER easy guide on Cerium element.
In fact, the table mentioned below is the perfect information box (Which gives you every single detail about the Cerium element in Periodic table.)
So if you want to know anything about Cerium element, then this guide is for you.
Let’s finish this very quickly.
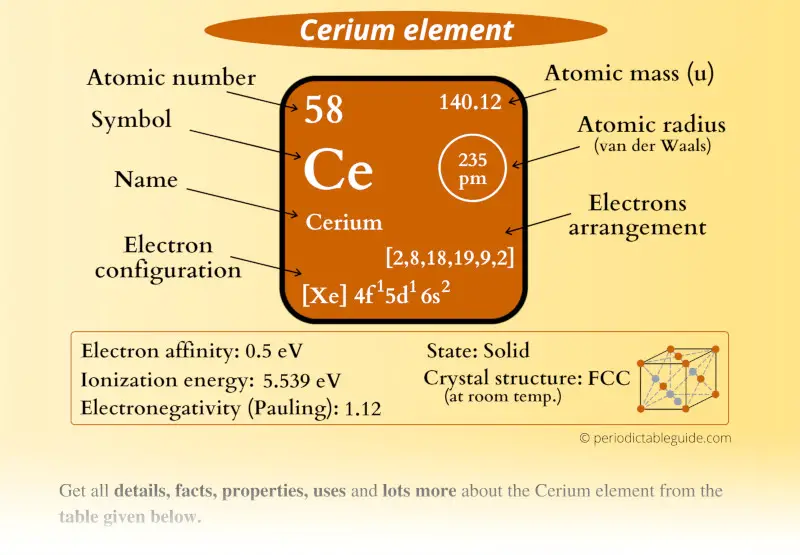
Cerium Element (Ce) Information
| Appearance |  Silvery white |
| State (at STP) | Solid |
| Position in Periodic table | 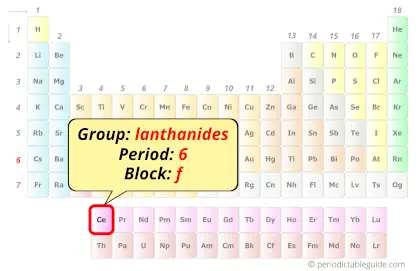 Group: lanthanides, Period: 6, Block: f |
| Category | 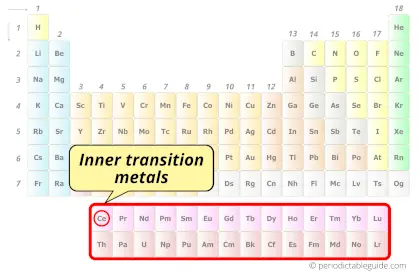 Inner transition metals |
| Atomic number or Protons | 58 |
| Neutrons | 82 |
| Electrons | 58 |
| Symbol | Ce |
| Atomic mass | 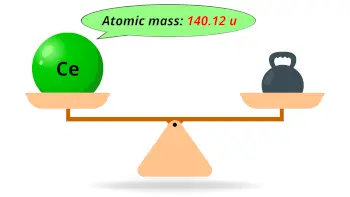 140.12 u |
| Electrons arrangement or Bohr model | 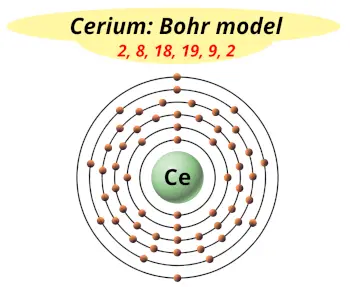 2, 8, 18, 19, 9, 2 |
| Electronic configuration | [Xe] 4f1 5d1 6s2 |
| Atomic radius | 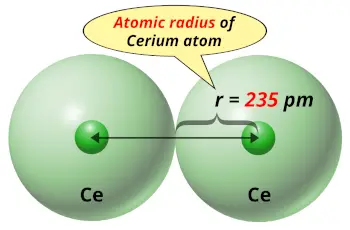 235 picometers (van der Waals radius) |
| 1st Ionization energy | 5.539 eV |
| Electronegativity | 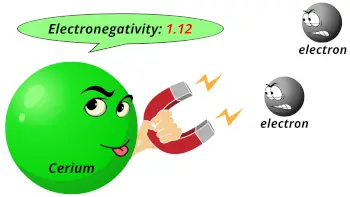 1.12 (Pauling scale) |
| Crystal structure | 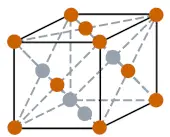 FCC (Face centered cubic) at room temperature |
| Melting point | 1068 K or 795 °C or 1463 °F |
| Boiling point | 3716 K or 3443 °C or 6229 °F |
| Density | 6.69 g/cm3 |
| Main isotope | 140Ce |
| Who discovered Cerium and when? | Wilhelm Hisinger, Jöns Jakob Berzelius, Martin Heinrich Klaproth (in 1803) |
| CAS number | 7440-45-1 |
Cerium in Periodic table
Cerium element is in period 6 and in lanthanide group of the Periodic table. Cerium is the f-block element and it belongs to inner transition metals group.
| H | He | ||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La* | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac** | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| *Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | ||||
| **Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
←Move to: Lanthanum (La) element – Periodic Table
→Move to: Praseodymium (Pr) element – Periodic Table
Why is Cerium in Period 6?
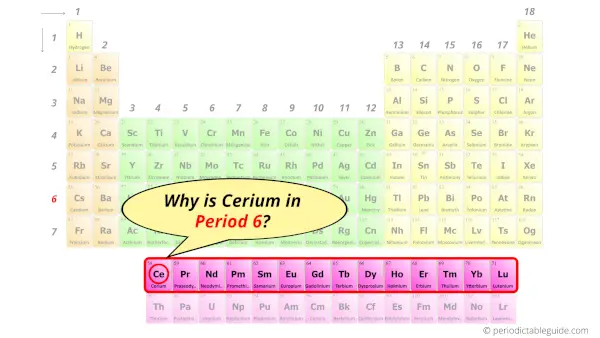
Let me ask you a question.
How many shells does cerium have?
It’s 6. Right?
You have already seen the bohr model of cerium atom in the above table.
From the Bohr model, it can be found that the number of orbits or shells in cerium is 6. Hence, as cerium has 6 orbits, it lies in period 6 of the Periodic table.
Why is Cerium in f-block?
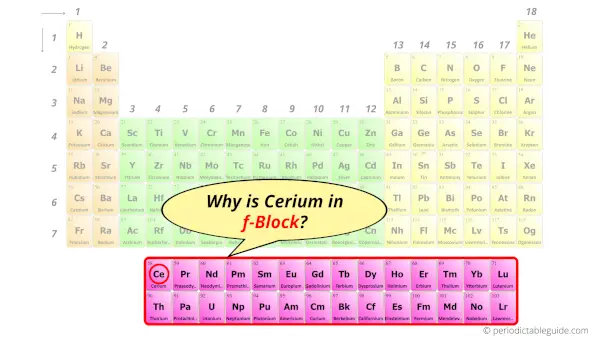
Before knowing this reason, first of all I want to ask you a simple question.
How can you determine the blocks-wise position of elements?
The simple answer: The elements will lie in the s, p, d or f block will completely depend upon the subshell in which the last electron will enter.
For example; the electron configuration of cerium is [Xe] 4f1 6s2 5d1.
So the last electron of cerium enters the f-subshell or f-orbital.
Hence, cerium is the f-block element.
Interesting facts about Cerium
Interesting facts about cerium element are mentioned below.
- The name Cerium was derived from the name of a dwarf planet “Ceres”.
- The cerium element was discovered by Wilhelm Hisinger & Jöns Jakob Berzelius and it was also individually discovered by another chemist Martin Heinrich Klaproth. Both of them discovered this element in the year 1803.
- India, Brazil and California are the three regions where the deposits of cerium are more.
- The proportion of cerium by weight is approximately 60 ppm in the earth’s crust.
- Cerium is a rare earth metal, but it is not actually rare in quantity. The fact is that it is spread evenly on the earth and it is very difficult to find this element at one place on the earth. Thus cerium is rare in the context of available resources.
Properties of Cerium
The physical and chemical properties of cerium element are mentioned below.
Physical properties of Cerium
Physical properties of cerium are mentioned below.
- Cerium is the solid metal having a Silvery white appearance.
- Cerium is a malleable metal which means it can be drawn into thin sheets.
- The melting point of cerium is 795 °C and its boiling point is 3443 °C.
- The atomic mass of cerium is 140.12 u and its density is 6.69 g/cm3.
- The crystal structure of cerium at room temperature is FCC (Face centered cubic).
- There are many isotopes of cerium, but out of them the most abundant isotope is 140Ce (its abundance is approximately 88.5%).
Chemical properties of Cerium
Chemical properties of cerium are mentioned below.
- Cerium is a chemically reactive rare earth metal. And hence it does not exist in its free state. It is always found in a compound form from the earth’s crust.
- When cerium is kept open in the air, it reacts with the oxygen of the air and forms oxides.
- Cerium metal also reacts with the concentrated acids as well as dilute acids.
- The first ionization energy of cerium is 5.539 eV.
- The electronegativity of cerium is 1.12 on the Pauling scale.
Uses of Cerium
Uses of cerium are mentioned below.
- Cigarette lighters use an alloy that contains cerium in it.
- Cerium is also used in carbon arc lighting that is used in studio lighting.
- Cerium is also used in catalytic converters that are used to treat the emissions common from exhaust of automobiles.
- Cerium oxide is used as an additive in diesel fuel which improves the engine performance.
Explore our New Interactive Periodic Table (with Rotating Bohr Models and More)

Details about this Periodic table:
- Access detailed info on all elements: atomic mass, electron configurations, charges, and more.
- View rotating Bohr models for all 118 elements.
- Get a free HD image of the Periodic Table.
Note: For future use, bookmark this Periodic table or visit “PeriodicTableGuide.com”
External resources:
- Cerium – Element information, properties and uses | Periodic Table. (n.d.). Cerium – Element Information, Properties and Uses | Periodic Table. https://www.rsc.org/periodic-table/element/58/cerium
- Cerium – Wikipedia. (2016, July 9). Cerium – Wikipedia. https://en.wikipedia.org/wiki/Cerium
- P. (n.d.). Cerium | Ce (Element) – PubChem. Cerium | Ce (Element) – PubChem. https://pubchem.ncbi.nlm.nih.gov/element/Cerium
- It’s Elemental – The Element Cerium. (n.d.). It’s Elemental – the Element Cerium. https://education.jlab.org/itselemental/ele058.html
- Atomic Data for Cerium (Ce). (n.d.). Atomic Data for Cerium (Ce). https://physics.nist.gov/PhysRefData/Handbook/Tables/ceriumtable1.htm
