
This is a SUPER easy guide on Oganesson element.
In fact, the table mentioned below is the perfect information box (Which gives you every single detail about the Oganesson element in Periodic table.)
So if you want to know anything about Oganesson element, then this guide is for you.
Let’s dive right into it!!!!
Oganesson Element (Og) Information
| State (at STP) | Gas (predicted) |
| Position in Periodic table | 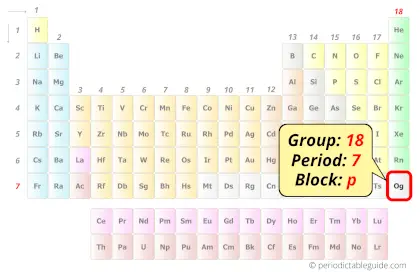 Group: 18, Period: 7, Block: p |
| Category | Synthetic element |
| Atomic number or Protons | 118 |
| Electrons | 118 |
| Symbol | Og |
| Atomic mass of Oganesson (most stable isotope) | 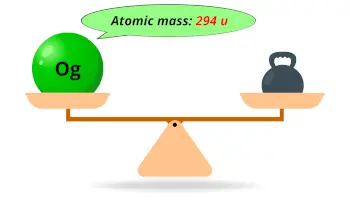 294 u |
| Electrons arrangement or Bohr model | 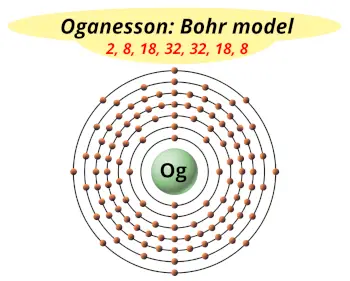 2, 8, 18, 32, 32, 18, 8 |
| Electronic configuration | [Rn] 5f14 6d10 7s2 7p6 |
| Crystal structure (predicted) | 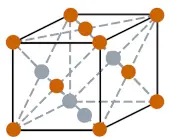 FCC (face centered cubic) |
| Density | 6.6-7.4 g/cm3 |
| Main isotope | 294Og |
| CAS number | 54144-19-3 |
Oganesson in Periodic table
Oganesson element is in group 18 and period 7 of the Periodic table. Oganesson is the p-block element and it is a radioactive synthetic element.
| H | He | ||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La* | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac** | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| *Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | ||||
| **Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
←Move to: Tennessine (Ts) element – Periodic Table
Why is Oganesson in Group 18?

Do you know, how many electrons can be accommodated in the first shell, second shell, third shell, fourth shell, etc…?
Here is the table showing the capacity of orbits to hold electrons.
Number of electrons in shells.
| Orbit / Shell (n) | Maximum no. of electrons this orbit can hold (2 × n2) |
| K shell, n = 1 | 2 × 1² = 2 |
| L shell, n = 2 | 2 × 2² = 8 |
| M shell, n = 3 | 2 × 3² = 18 |
| N shell, n = 4 | 2 × 4² = 32 |
| . . . | . . . |
Thus,
- 1st shell can hold 2 electrons.
- 2nd shell can hold 8 electrons.
- 3rd shell can hold 18 electrons.
- 4th shell can hold 32 electrons, and so on…
Now the atomic number of Oganesson (Og) is 118.
Hence the Oganesson element has electron arrangement 2, 8, 18, 32, 32, 18, 8.
This electron arrangement indicates that the outermost orbit of oganesson element (Og) has 8 electrons.
Hence, it lies in group 18.
Why is Oganesson in Period 7?
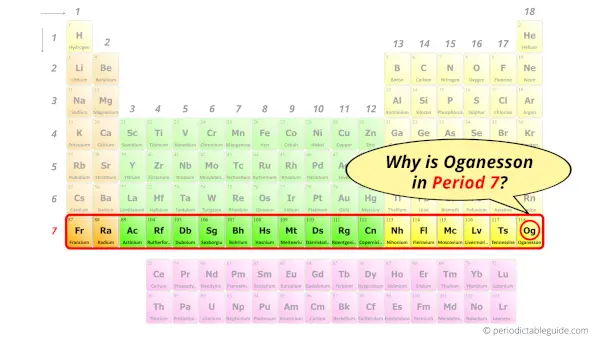
Let me ask you a question.
How many shells does an Oganesson atom have?
It’s 7. Right?
You have already seen the bohr model of oganesson atom in the above table.
From the Bohr model, it can be found that the number of orbits or shells in oganesson is 7. Hence, as oganesson has 7 orbits, it lies in period 7 of the Periodic table.
5 facts about Oganesson
Interesting facts about oganesson element are mentioned below.
- The element was given the name “Oganesson” to honor the physicist “Yuri Oganessian”.
- Oganesson is highly radioactive and it is not available naturally.
- The Oganesson element has the highest atomic mass of all the known elements of periodic table. Its predicted atomic mass is 294 u.
- The oganesson element was discovered by the scientists at Lawrence Livermore National laboratory (California) and Joint Institute for Nuclear Research (Russia) in 2002.
- There are 5 isotopes of oganesson detected till now. And all these isotopes are radioactive in nature.
- The longest lived isotope of oganesson is 294Og, and its half life is 0.69 milliseconds.
Properties of Oganesson
The physical and chemical properties of oganesson element are mentioned below.
- Oganesson is extremely radioactive and it has a very very short half life (of just a few milliseconds).
- As a group 18 element, it is predicted to have complete octet in the outermost shell.
- The predicted atomic mass of oganesson is 294 u.
- The common oxidation states predicted for oganesson are +2 and +4.
- The predicted crystal structure of Oganesson is FCC (i.e face centered cubic).
Uses of Oganesson
Oganesson is basically used for scientific research work. Oganesson has no commercial use due to its short life and expensive production.
Explore our New Interactive Periodic Table (with Rotating Bohr Models and More)

Details about this Periodic table:
- Access detailed info on all elements: atomic mass, electron configurations, charges, and more.
- View rotating Bohr models for all 118 elements.
- Get a free HD image of the Periodic Table.
Note: For future use, bookmark this Periodic table or visit “PeriodicTableGuide.com”
External resources:
- Oganesson – Wikipedia. (2017, December 19). Oganesson – Wikipedia. https://en.wikipedia.org/wiki/Oganesson
- Oganesson – Element information, properties and uses | Periodic Table. (n.d.). Oganesson – Element Information, Properties and Uses | Periodic Table. https://www.rsc.org/periodic-table/element/118/oganesson
- P. (n.d.). Oganesson | Og (Element) – PubChem. Oganesson | Og (Element) – PubChem. https://pubchem.ncbi.nlm.nih.gov/element/Oganesson
- Oganesson – American Chemical Society. (2020, October 5). American Chemical Society. https:///molecule-of-the-week/archive/o/oganesson.html
- It’s Elemental – The Element Oganesson. (n.d.). It’s Elemental – the Element Oganesson. https://education.jlab.org/itselemental/ele118.html
- Periodic Table of Elements: Los Alamos National Laboratory. (n.d.). Periodic Table of Elements: Los Alamos National Laboratory. https://periodic.lanl.gov/118.shtml
- Oganesson – noble but not a gas. (2018, February 8). MSUToday | Michigan State University. https://msutoday.msu.edu/news/2018/oganesson-noble-but-not-a-gas
