This is a SUPER easy guide on Roentgenium element.
In fact, the table mentioned below is the perfect information box (Which gives you every single detail about the Roentgenium element in Periodic table.)
So if you want to know anything about Roentgenium element, then this guide is for you.
Let’s dive right into it!
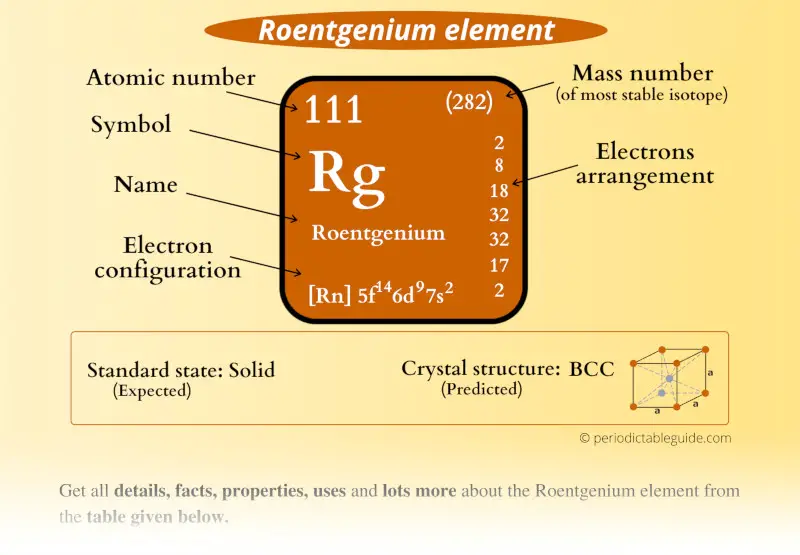
Roentgenium Element (Rg) Information
| Appearance | Silvery (predicted) |
| State (at STP) | Solid (predicted) |
| Position in Periodic table | 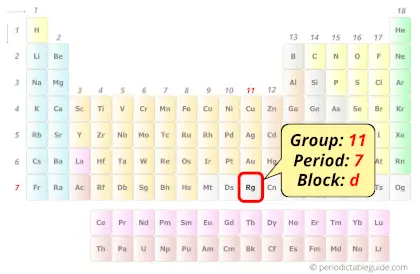 Group: 11, Period: 7, Block: d |
| Category | Synthetic element |
| Atomic number or Protons | 111 |
| Neutrons | 161 |
| Electrons | 111 |
| Symbol | Rg |
| Atomic mass of Roentgenium (most stable isotope) | 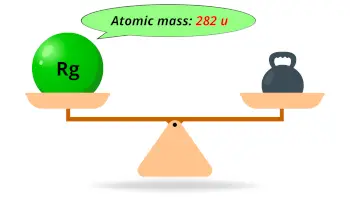 282 u |
| Electrons arrangement or Bohr model | 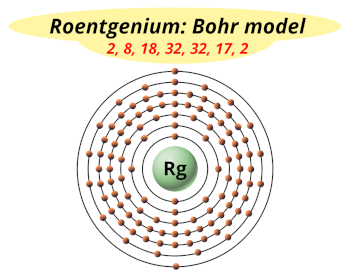 2, 8, 18, 32, 32, 17, 2 |
| Electronic configuration | [Rn] 5f14 6d9 7s2 |
| Crystal structure (predicted) | 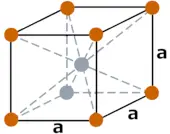 BCC (body centered cubic) |
| Density | 22-24 g/cm3 |
| CAS number | 54386-24-2 |
Roentgenium in Periodic table
Roentgenium element is in group 11 and in period 7 of the Periodic table. Roentgenium is the d-block element and it is a radioactive synthetic element.
| H | He | ||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La* | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac** | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| *Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | ||||
| **Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
←Move to: Darmstadtium (Ds) element – Periodic Table
→Move to: Copernicium (Cn) element – Periodic Table
Why is Roentgenium in Period 7?
Let me ask you a question.
How many shells does a roentgenium atom have?
It’s 7. Right?
You have already seen the bohr model of roentgenium atom in the above table.
From the Bohr model, it can be found that the number of orbits or shells in roentgenium is 7. Hence, as roentgenium has 7 orbits, it lies in period 7 of the Periodic table.
Why is Roentgenium in d-block?
Before knowing this reason, first of all I want to ask you a simple question.
How can you determine the blocks-wise position of elements?
The simple answer: The elements will lie in the s, p, d or f block will completely depend upon the subshell in which the last electron will enter.
For example; the electron configuration of roentgenium is [Rn] 5f14 6d9 7s2.
So the last electron of roentgenium enters the d-subshell or d-orbital.
Hence, roentgenium is the d-block element.
5 facts about Roentgenium
Interesting facts about roentgenium element are mentioned below.
- The element was given the name “Roentgenium” to honor the physicist Wilhelm Rontgen.
- Roentgenium was discovered by Peter Armbruster and Gottfried Münzenberg in 1994.
- Roentgenium is artificially prepared in a laboratory and it is not available naturally.
- There are 9 known isotopes of roentgenium and all those isotopes are radioactive in nature.
- The most stable isotope of roentgenium is 282Rg, which has a half-life of only 100 seconds.
Properties of Roentgenium
The physical and chemical properties of roentgenium element are mentioned below.
- The roentgenium element is extremely radioactive and has a very short half life.
- The calculated atomic mass of the most stable isotope of roentgenium is 282 u and its predicted density is 22-24 g/cm3.
- The predicted phase of roentgenium is solid at STP.
- The predicted crystal structure of roentgenium is BCC (body centered cubic).
- The most stable oxidation state of roentgenium is predicted to be +3. Besides this, it can also show other oxidation states like +1, +5 and +7.
Uses of Roentgenium
Roentgenium is generally used for scientific research work in chemistry. There is no commercial use of roentgenium due to its radioactive nature and scarcity.
Explore our New Interactive Periodic Table (with Rotating Bohr Models and More)

Details about this Periodic table:
- Access detailed info on all elements: atomic mass, electron configurations, charges, and more.
- View rotating Bohr models for all 118 elements.
- Get a free HD image of the Periodic Table.
Note: For future use, bookmark this Periodic table or visit “PeriodicTableGuide.com”
External resources:
- Roentgenium – Wikipedia. (2013, October 4). Roentgenium – Wikipedia. https://en.wikipedia.org/wiki/Roentgenium
- Roentgenium – Element information, properties and uses | Periodic Table. (n.d.). Roentgenium – Element Information, Properties and Uses | Periodic Table. https://www.rsc.org/periodic-table/element/111/roentgenium
- P. (n.d.). Roentgenium | Rg (Element) – PubChem. Roentgenium | Rg (Element) – PubChem. https://pubchem.ncbi.nlm.nih.gov/element/Roentgenium
- It’s Elemental – The Element Roentgenium. (n.d.). It’s Elemental – the Element Roentgenium. https://education.jlab.org/itselemental/ele111.html
