
This is a SUPER easy guide on Gadolinium element.
In fact, the table mentioned below is the perfect information box (Which gives you every single detail about the Gadolinium element in Periodic table.)
So if you want to know anything about the Gadolinium element, then this guide is for you.
Let’s dive right into it!
Gadolinium Element (Gd) Information
| Appearance |  Silvery white metallic appearance |
| State (at STP) | Solid |
| Position in Periodic table | 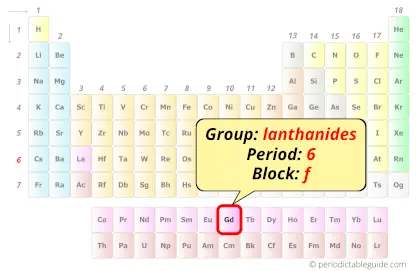 Group: lanthanides, Period: 6, Block: f |
| Category | 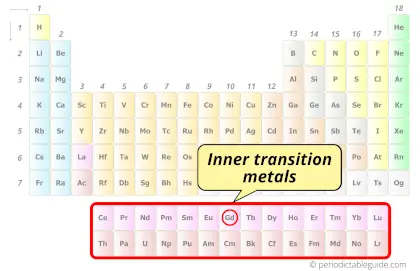 Inner transition metals |
| Atomic number or Protons | 64 |
| Neutrons | 93 |
| Electrons | 64 |
| Symbol | Gd |
| Atomic mass | 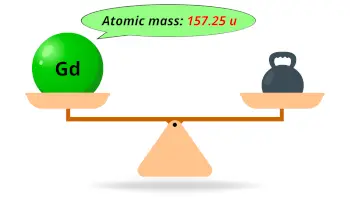 157.25 u |
| Electrons arrangement or Bohr model | 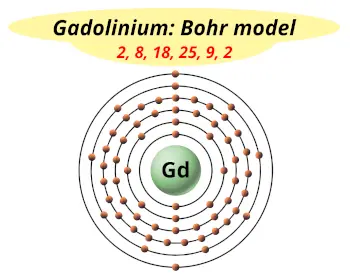 2, 8, 18, 25, 9, 2 |
| Electronic configuration | [Xe] 4f7 5d1 6s2 |
| Atomic radius | 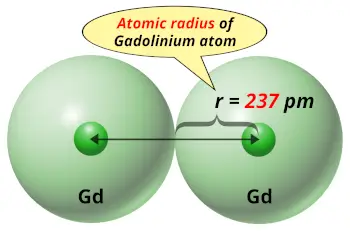 237 picometers (van der Waals radius) |
| 1st Ionization energy | 6.150 eV |
| Electronegativity | 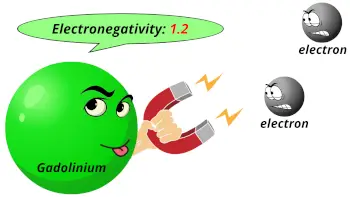 1.2 (Pauling scale) |
| Crystal structure | 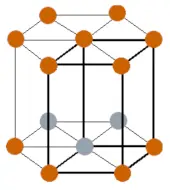 HCP (hexagonal close packed) |
| Melting point | 1585 K or 1312 °C or 2394 °F |
| Boiling point | 3273 K or 3000 °C or 5432 °F |
| Density | 7.9 g/cm3 |
| Main isotope | 158Gd |
| Who discovered Gadolinium and when? |  Jean Charles Galissard de Marignac in 1880 |
| CAS number | 7440-54-2 |
Gadolinium in Periodic table
Gadolinium element is in period 6 and in lanthanide group of the Periodic table. Gadolinium is the f-block element and it belongs to inner transition metals group.
| H | He | ||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La* | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac** | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| *Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | ||||
| **Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
←Move to: Europium (Eu) element – Periodic Table
→Move to: Terbium (Tb) element – Periodic Table
Why is Gadolinium in Period 6?
Let me ask you a question.
How many shells does gadolinium have?
It’s 6. Right?
You have already seen the bohr model of gadolinium atom in the above table.
From the Bohr model, it can be found that the number of orbits or shells in gadolinium is 6. Hence, as gadolinium has 6 orbits, it lies in period 6 of the Periodic table.
Why is Gadolinium in f-block?

Before knowing this reason, first of all I want to ask you a simple question.
How can you determine the blocks-wise position of elements?
The simple answer: The elements will lie in the s, p, d or f block will completely depend upon the subshell in which the last electron will enter.
For example; the electron configuration of gadolinium is [Xe] 4f7 5d1 6s2.
So the last electron of gadolinium enters the f-subshell or f-orbital.
Hence, gadolinium is the f-block element.
5 Interesting facts about Gadolinium
Interesting facts about gadolinium element are mentioned below.
- The concentration of gadolinium in the earth’s crust is approximately 5.2 ppm by weight.
- Gadolinium was discovered by Jean Charles Galissard de Marignac in 1880. Later on it was first isolated by Lecoq de Boisbaudran in 1886.
- From the earth’s crust, gadolinium is mostly obtained from the mineral gadolinite.
- Gadolinium is a rare earth metal, but it is not actually rare in quantity. The fact is that it is spread evenly on the earth and it is very difficult to find this element at one place on the earth. Thus it is rare in the context of available resources.
- Out of all the elements of periodic table, gadolinium element has the greatest ability to absorb the thermal neutrons.
Properties of Gadolinium
The physical and chemical properties of gadolinium element are mentioned below.
Physical properties of Gadolinium
Physical properties of gadolinium are mentioned below.
- Gadolinium is a ductile and malleable metal having a silvery white metallic appearance.
- The melting point of gadolinium is 1312 °C and its boiling point is 3000 °C.
- The crustal structure of gadolinium at room temperature is HCP (hexagonal close packed).
- Gadolinium has many isotopes, and out of these isotopes, the most abundant isotope is 158Gd which has an abundance of approximately 24.8 %.
Chemical properties of Gadolinium
Chemical properties of gadolinium are mentioned below.
- Due to the reactivity of gadolinium , it is not found in its free state. But it is always found as a compound with other elements in the earth’s crust.
- When gadolinium is kept open in the moist air, it starts tarnishing.
- Gadolinium also shows a chemical reaction with water.
- Gadolinium is a rare earth metal that is soluble in dilute acids.
- The most common oxidation state of gadolinium is +3, which is also common for other rare earth elements.
Uses of Gadolinium
Uses of gadolinium are mentioned below.
- Phosphors used in color TV picture tubes contain some proportion of gadolinium element in it.
- Gadolinium is also added to iron and chromium to improve its oxidation as well as its workability.
- The compounds of Gadolinium are also used in microwave applications.
- Gadolinium also has a capacity to absorb neutrons, hence it is used in nuclear reactors for absorbing neutrons. [1]
Explore our New Interactive Periodic Table (with Rotating Bohr Models and More)

Details about this Periodic table:
- Access detailed info on all elements: atomic mass, electron configurations, charges, and more.
- View rotating Bohr models for all 118 elements.
- Get a free HD image of the Periodic Table.
Note: For future use, bookmark this Periodic table or visit “PeriodicTableGuide.com”
External resources:
- Gadolinium – Element information, properties and uses | Periodic Table. (n.d.). Gadolinium – Element Information, Properties and Uses | Periodic Table. https://www.rsc.org/periodic-table/element/64/gadolinium
- Gadolinium – Wikipedia. (2009, June 6). Gadolinium – Wikipedia. https://en.wikipedia.org/wiki/Gadolinium
- Gadolinium | chemical element. (n.d.). Encyclopedia Britannica. https://www.britannica.com/science/gadolinium
- P. (n.d.). Gadolinium | Gd (Element) – PubChem. Gadolinium | Gd (Element) – PubChem. https://pubchem.ncbi.nlm.nih.gov/element/Gadolinium
- It’s Elemental – The Element Gadolinium. (n.d.). It’s Elemental – the Element Gadolinium. https://education.jlab.org/itselemental/ele064.html
- Periodic Table of Elements: Los Alamos National Laboratory. (n.d.). Periodic Table of Elements: Los Alamos National Laboratory. https://periodic.lanl.gov/64.shtml
