This is a SUPER easy guide on Chlorine element.
In fact, the table mentioned below is the perfect information box (Which gives you every single detail about the Chlorine element in Periodic table.)
So if you want to know anything about Chlorine element, then this guide is for you.
Let’s dive right into it!
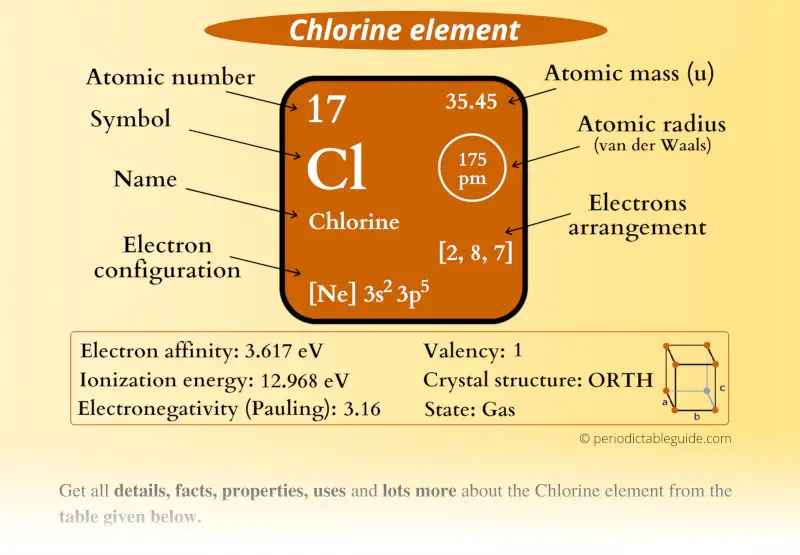
Chlorine Element (Cl) Information
| Appearance | Pale yellow-green gas |
| State (at STP) | Gas |
| Position in Periodic table | 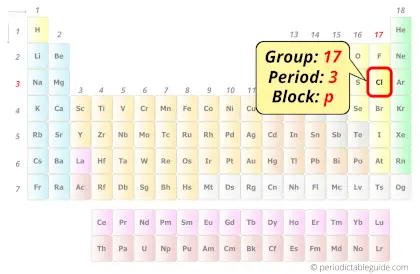 Group: 17, Period: 3, Block: p |
| Category | 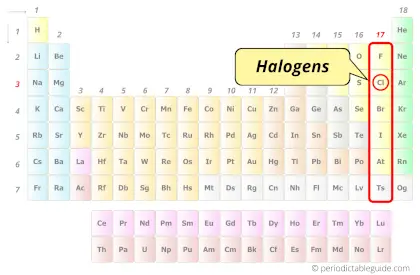 Halogens |
| Atomic number or Protons | 17 |
| Neutrons | 18 |
| Electrons | 17 |
| Symbol | Cl |
| Atomic mass | 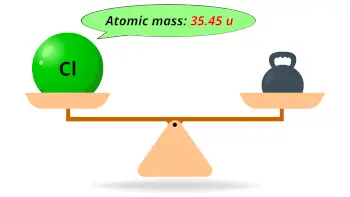 35.45 u |
| Electrons arrangement or Bohr model | 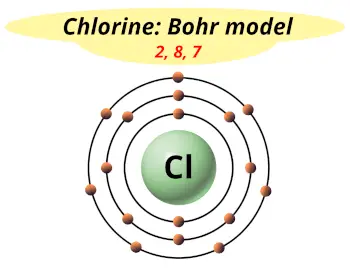 2, 8, 7 |
| Electronic configuration | [Ne] 3s2 3p5 |
| Atomic radius | 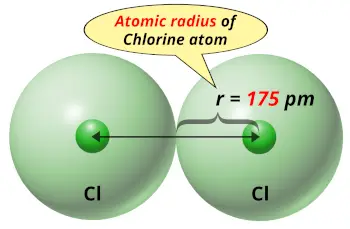 175 picometers (van der Waals radius) |
| Valence electrons | 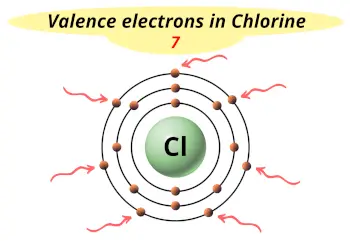 7 |
| 1st Ionization energy | 12.968 eV |
| Electronegativity | 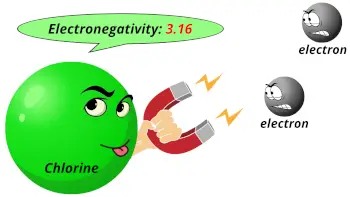 3.16 (Pauling scale) |
| Crystal structure | 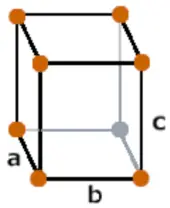 Orthorhombic |
| Melting point of Chlorine (Cl2) | 171.6 K or -101.5 °C or -105.7 °F |
| Boiling point of Chlorine (Cl2) | 239.11 K or -34.04 °C or -29.27 °F |
| Density | 3.214 g/L |
| Main isotopes | 35Cl and 37Cl |
| Who discovered Chlorine and when? |  Carl Wilhelm Scheele in 1774 |
| CAS number of (Cl2) | 7782-50-5 |
Chlorine in Periodic table
Chlorine element is in group 17 and period 3 of the Periodic table. Chlorine is the p-block element and it belongs to halogens group.
| H | He | ||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La* | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac** | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| *Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | ||||
| **Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
←Move to: Sulfur (S) element – Periodic Table
→Move to: Argon (Ar) element – Periodic Table
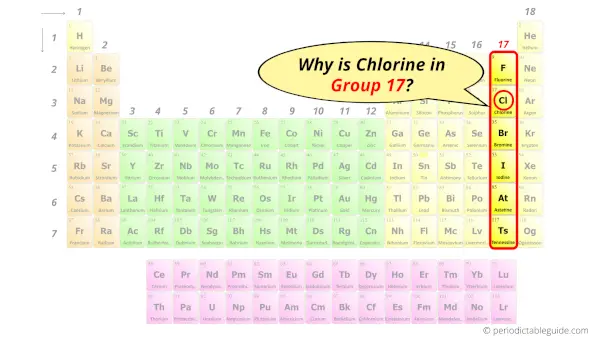
Why is Chlorine in Group 17?
Do you know, how many electrons can be accommodated in the first shell, second shell, third shell, fourth shell, etc…?
Here is the table showing the capacity of orbits to hold electrons.
Number of electrons in shells.
| Orbit / Shell (n) | Maximum no. of electrons this orbit can hold |
| K shell, n = 1 | 2 × 1² = 2 |
| L shell, n = 2 | 2 × 2² = 8 |
| M shell, n = 3 | 2 × 3² = 18 |
| N shell, n = 4 | 2 × 4² = 32 |
Thus,
- 1st shell can hold 2 electrons.
- 2nd shell can hold 8 electrons.
- 3rd shell can hold 18 electrons.
- 4th shell can hold 32 electrons.
Now the atomic number of chlorine (Cl) is 17.
Hence the chlorine element has electrons arrangement 2, 8, 7.
This electron arrangement indicates that the outermost orbit of chlorine element (Cl) has 7 electrons.
Hence, it lies in group 17.
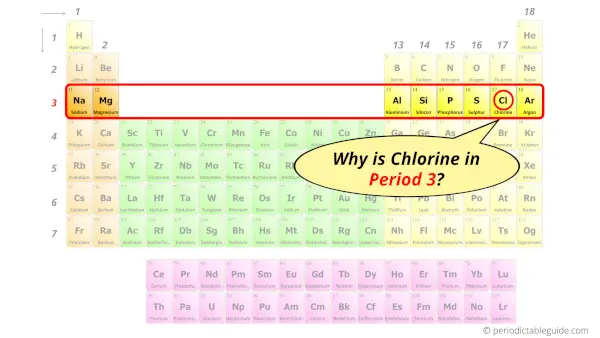
Why is Chlorine in Period 3?
Let me ask you a question.
How many shells does chlorine have?
It’s 3. Right?
You have already seen the bohr model of chlorine element in the above table.
From the Bohr model, it can be found that the number of orbits or shells in chlorine is 3. Hence, as chlorine has 3 orbits, it lies in period 3 of the Periodic table.
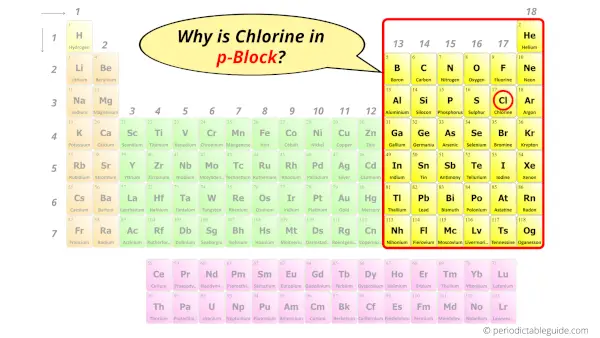
Why is Chlorine in p-block?
Before knowing this reason, first of all I want to ask you a simple question.
How can you determine the blocks-wise position of elements?
The simple answer: The elements will lie in the s, p, d or f block will completely depend upon the subshell in which the last electron will enter.
For example; the electron configuration of chlorine is [Ne] 3s2 3p5.
So the last electron of chlorine enters the p-subshell or p-orbital.
Hence, chlorine is the p-block element.
7 Interesting facts about Chlorine
Interesting facts about chlorine are mentioned below.
- The name “chlorine” was derived from the Greek word “khloros” which means “greenish-yellow” color. This is because chlorine exists as a greenish-yellow colored gas.
- Chlorine is always found as a diatomic molecule (i.e Cl2).
- The oceans of the earth contain an excess amount of chlorine (which is present in NaCl dissolved in water). This makes chlorine the 3rd most abundant element in the ocean.
- If we talk about the abundance of chlorine in the earth’s crust, then chlorine is the 21st most abundant element.
- Human body also contains chlorine element.
- Chlorine is the 2nd lightest halogen present on the periodic table (the lightest halogen is fluorine).
- Human body intake chlorine from table salt (i.e NaCl) but inhaling pure chlorine gas is very toxic.
Properties of Chlorine
The physical and chemical properties of chlorine element are mentioned below.
Physical properties of Chlorine
Physical properties of chlorine are mentioned below.
- Chlorine is a pale yellow-green colored gas having a strong smell.
- The melting point of diatomic chlorine gas (Cl2) is -101.5 °C and it’s boiling point is -34.04 °C.
- Density of chlorine gas is 3.214 g/L which is far more than the density of the atmospheric gases of the earth.
Chemical properties of Chlorine
Chemical properties of chlorine are mentioned below.
- Chlorine is chemically reactive and hence it is not found in a free state, but it is always found as a compound with other elements (for example, NaCl, HCl, etc).
- Chlorine has high electronegativity and higher electron affinity, so it reacts with almost all the elements except with lighter noble gases.
- Chlorine is a non explosive and non flammable gas.
- Chlorine reacts with ammonia and forms white mist. Hence the leakage of chlorine gas from the containers can be detected by using ammonia in this way.
- Chlorine is also used to make drinking water safe from microorganisms.
- Electrolysis of salt water can produce chlorine gas.
- The chlorine present in CFC destroys the ozone molecules and hence it should be handled very carefully.
Uses of Chlorine
Uses of chlorine are mentioned below.
- In earlier days, chlorine was used as a chemical weapon as it can burn the skin.
- Chlorine element is used in manufacturing of paper, plastics, textiles, dies, etc.
- It is also used in manufacturing of medicines, disinfectants as well as insecticides.
- Chlorine is added to the swimming pool water to keep it clean.
- PVC (polyvinyl chloride) is the highly used polymer which contains chlorine in it.
Explore our New Interactive Periodic Table (with Rotating Bohr Models and More)

Details about this Periodic table:
- Access detailed info on all elements: atomic mass, electron configurations, charges, and more.
- View rotating Bohr models for all 118 elements.
- Get a free HD image of the Periodic Table.
Note: For future use, bookmark this Periodic table or visit “PeriodicTableGuide.com”
External resources:
- Chlorine. (2022, April 5). American Chemistry Council. https://www.americanchemistry.com/industry-groups/chlorine
- The Facts About Chlorine. (2005, May 1). The Facts About Chlorine. https://www.health.ny.gov/environmental/emergency/chemical_terrorism/chlorine_tech.htm
- C&EN: IT’S ELEMENTAL: THE PERIODIC TABLE – CHLORINE. (n.d.). C&EN: IT’S ELEMENTAL: THE PERIODIC TABLE – CHLORINE. https://pubsapp.acs.org/cen/80th/print/chlorine.html?
- Chlorine – Element information, properties and uses | Periodic Table. (n.d.). Chlorine – Element Information, Properties and Uses | Periodic Table. https://www.rsc.org/periodic-table/element/17/chlorine
- Chlorine – Wikipedia. (2008, July 10). Chlorine – Wikipedia. https://en.wikipedia.org/wiki/Chlorine
