This is a SUPER easy guide on Argon element.
In fact, the table mentioned below is the perfect information box (Which gives you every single detail about the Argon element in Periodic table.)
So if you want to know anything about the Argon element, then this guide is for you.
Let’s finish this very quickly.
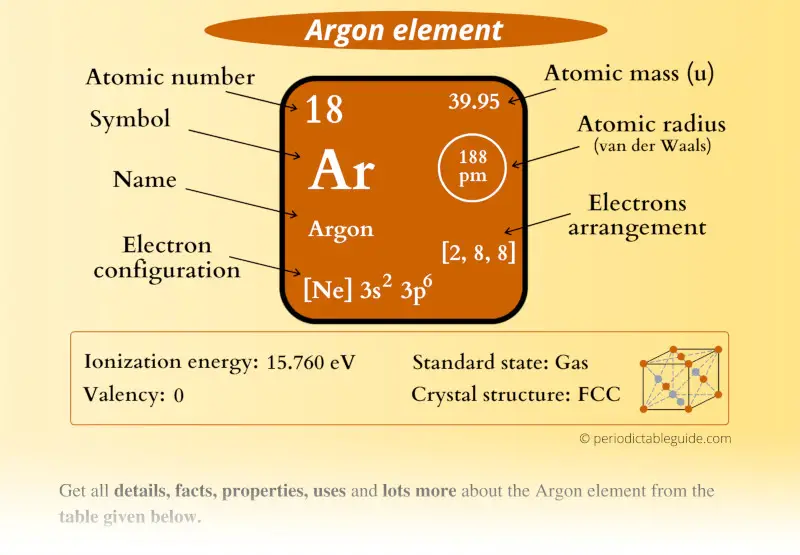
Argon Element (Ar) Information
| Appearance | Colorless gas |
| State (at STP) | Gas |
| Position in Periodic table | 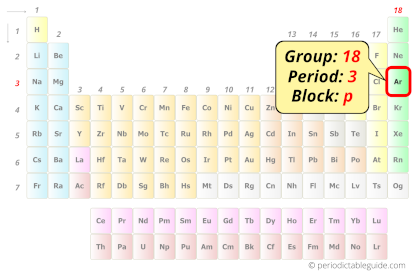 Group: 18, Period: 3, Block: p |
| Category | 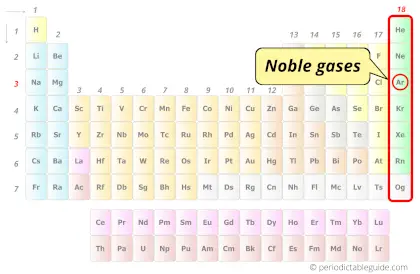 Noble gases |
| Atomic number or Protons | 18 |
| Neutrons | 22 |
| Electrons | 18 |
| Symbol | Ar |
| Atomic mass | 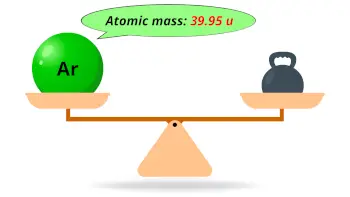 39.95 u |
| Electrons arrangement or Bohr model | 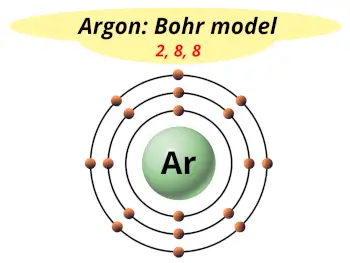 2, 8, 8 |
| Electronic configuration | [Ne] 3s2 3p6 |
| Atomic radius | 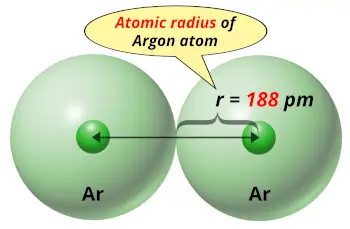 188 picometers (van der Waals radius) |
| Valence electrons | 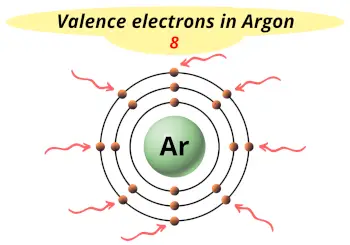 8 |
| 1st Ionization energy | 15.76 eV |
| Crystal structure | 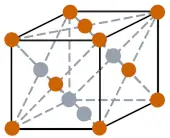 FCC (Face-centered cubic) |
| Melting point | 83.81 K or -189.34 °C or -308.81 °F |
| Boiling point | 87.3 K or -185.84 °C or -302.52 °F |
| Density | 1.79 g/L |
| Main isotope | 40Ar |
| Who discovered Argon and when? |  Lord Rayleigh and William Ramsay in 1894 |
| CAS number | 7440-37-1 |
Argon in Periodic table
Argon element is in group 18 and period 3 of the Periodic table. Argon is the p-block element and it belongs to Noble gases group.
| H | He | ||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La* | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac** | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| *Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | ||||
| **Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
←Move to: Chlorine (Cl) element – Periodic Table
→Move to: Potassium (K) element – Periodic Table
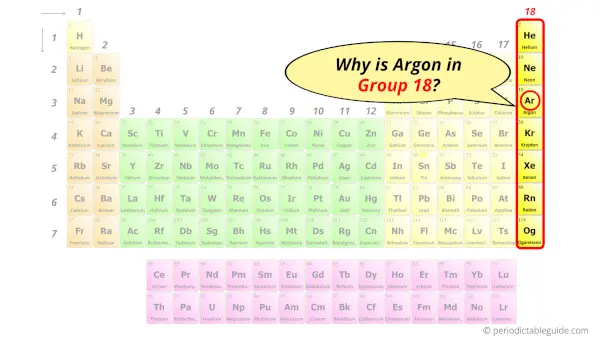
Why is Argon in Group 18?
Do you know, how many electrons can be accommodated in the first shell, second shell, third shell, fourth shell, etc…?
Here is the table showing the capacity of orbits to hold electrons.
Number of electrons in shells.
| Orbit / Shell (n) | Maximum no. of electrons this orbit can hold |
| K shell, n = 1 | 2 × 1² = 2 |
| L shell, n = 2 | 2 × 2² = 8 |
| M shell, n = 3 | 2 × 3² = 18 |
| N shell, n = 4 | 2 × 4² = 32 |
Thus,
- 1st shell can hold 2 electrons.
- 2nd shell can hold 8 electrons.
- 3rd shell can hold 18 electrons.
- 4th shell can hold 32 electrons.
Now the atomic number of Argon (Ar) is 18.
Hence the argon element has electrons arrangement 2, 8, 8.
This electron arrangement indicates that the outermost orbit of argon element (Ar) has 8 electrons.
Hence, it lies in group 18.
Why is Argon in Period 3?

Let me ask you a question.
How many shells does argon have?
It’s 3. Right?
You have already seen the bohr model of argon element in the above table.
From the Bohr model, it can be found that the number of orbits or shells in argon is 3. Hence, as argon has 3 orbits, it lies in period 3 of the Periodic table.
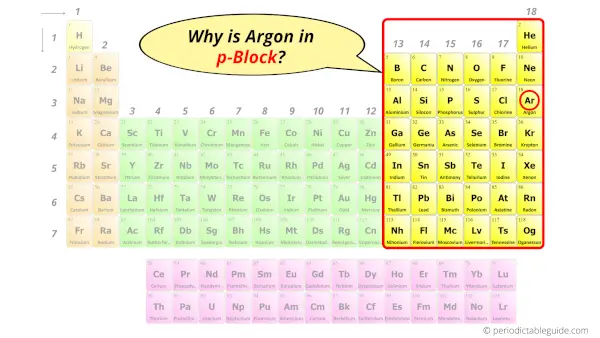
Why is Argon in p-block?
Before knowing this reason, first of all I want to ask you a simple question.
How can you determine the blocks-wise position of elements?
The simple answer: The elements will lie in the s, p, d or f block will completely depend upon the subshell in which the last electron will enter.
For example; the electron configuration of argon is [Ne] 3s2 3p6.
So the last electron of argon enters the p-subshell or p-orbital.
Hence, argon is the p-block element.
7 Interesting facts about Argon
Interesting facts about the argon element are mentioned below.
- The amount of argon present in the earth’s atmosphere is only 0.94% by volume.
- The 3rd most abundant gas found in the earth’s atmosphere is argon.
- The symbol of argon used to be A, but later on in 1957, IUPAC changed its symbol to Ar.
- Out of all the noble gases present on the periodic table, argon is the noble gas that was discovered first.
- The name “argon” was derived from the Greek word “argos”, which means inactive.
- On the earth, the manufacturing of argon is done by radioactive decay of potassium, but in space, argon is made in stars.
- Argon is denser than air, so it remains close to the earth’s surface when it is used.
Properties of Argon
The physical and chemical properties of argon element are mentioned below.
Physical properties of Argon
Physical properties of argon are mentioned below.
- Argon is a colorless, odourless and tasteless gas.
- In liquid state also, the argon remains colorless.
- When argon is ionized, it glows with violet light.
- Argon has many isotopes, but out of those isotopes, 40Ar has an abundance of 99.6%.
- Density of argon is 1.79 g/L which is 38% more than the density of air.
- The melting point of argon is -189.34 °C and its boiling point is -185.84 °C.
- The van der Waals radius of argon is 188 picometers.
Chemical properties of Argon
Chemical properties of argon are mentioned below.
- Argon is chemically inert gas, and so it does not react with any other elements. Because of this reason, argon is classified as inert gas.
- Argon is an inert gas, but it has been found that argon forms a compound argon fluorohydride at temperature below -256.15 °C.
Uses of Argon
Uses of argon are mentioned below.
- Argon gas is used to provide an inert atmosphere during various welding processes.
- Argon is widely used in fluorescent bulbs. It is also used in incandescent lights to protect the filament from oxygen.
- Argon is also used to store sensitive chemicals as well as it is also used to protect some materials.
- Argon is used to preserve important historical documents or maps. The argon gas is simply pumped over these materials which does not degrade the ink or paper of the document (or map).
- Argon is used to preserve the drinks by adding argon on the top of wine bottles. The argon is heavier than air, so it settles on the top surface of the wine (or any other drink), and this prevents the contact of wine with oxygen.
- Argon is also used in double glazed windows in the space between the two panes. This provides thermal insulation as the argon is inert gas.
Explore our New Interactive Periodic Table (with Rotating Bohr Models and More)

Details about this Periodic table:
- Access detailed info on all elements: atomic mass, electron configurations, charges, and more.
- View rotating Bohr models for all 118 elements.
- Get a free HD image of the Periodic Table.
Note: For future use, bookmark this Periodic table or visit “PeriodicTableGuide.com”
External resources:
- Periodic Table of Elements: Los Alamos National Laboratory. (n.d.). Periodic Table of Elements: Los Alamos National Laboratory. https://periodic.lanl.gov/18.shtml
- Argon – Element information, properties and uses | Periodic Table. (n.d.). Argon – Element Information, Properties and Uses | Periodic Table. https://www.rsc.org/periodic-table/element/18/argon
- It’s Elemental – The Element Argon. (n.d.). It’s Elemental – the Element Argon. https://education.jlab.org/itselemental/ele018.html
- P. (n.d.). Argon | Ar (Element) – PubChem. Argon | Ar (Element) – PubChem. https://pubchem.ncbi.nlm.nih.gov/element/Argon
