This is a SUPER easy guide on Promethium element.
In fact, the table mentioned below is the perfect information box (Which gives you every single detail about the Promethium element in Periodic table.)
So if you want to know anything about Promethium element, then this guide is for you.
Let’s finish this very quickly.
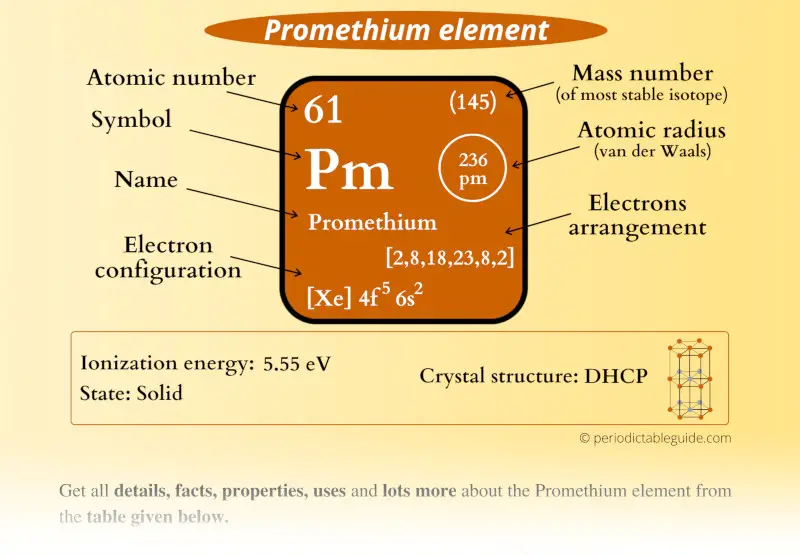
Promethium Element (Pm) Information
| Appearance | Metallic appearance |
| State (at STP) | Solid |
| Position in Periodic table |  Group: lanthanides, Period: 6, Block: f |
| Category |  Inner transition metals |
| Atomic number or Protons | 61 |
| Neutrons | 84 |
| Electrons | 61 |
| Symbol | Pm |
| Atomic mass | 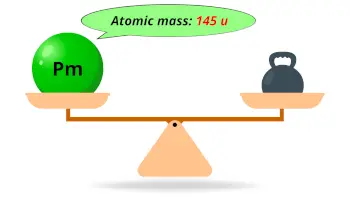 145 u |
| Electrons arrangement or Bohr model | 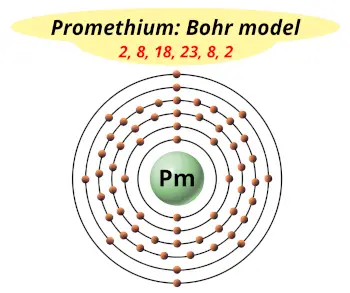 2, 8, 18, 23, 8, 2 |
| Electronic configuration | [Xe] 4f5 6s2 |
| Atomic radius | 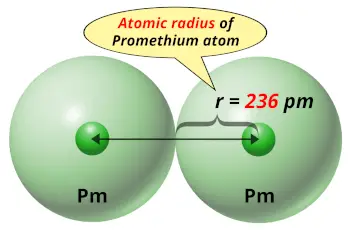 236 picometers (van der Waals radius) |
| 1st Ionization energy | 5.55 eV |
| Crystal structure | 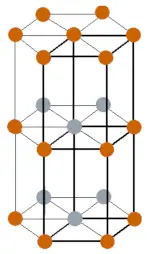 Double hexagonal close packed |
| Melting point | 1315 K or 1042 °C or 1908 °F |
| Boiling point | 3273 K or 3000 °C or 5432 °F |
| Density | 7.26 g/cm3 |
| Main isotope | 145Pm and 147Pm |
| Who discovered Promethium and when? | Lawrence Glendenin, Jacob Marinsky, Charles Coryell (in 1945) |
| CAS number | 7440-12-2 |
Promethium in Periodic table
Promethium element is in period 6 and in lanthanide group of the Periodic table. Promethium is the f-block element and it belongs to inner transition metals group.
| H | He | ||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La* | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac** | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| *Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | ||||
| **Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
←Move to: Neodymium (Nd) element – Periodic Table
→Move to: Samarium (Sm) element – Periodic Table
Why is Promethium in Period 6?
Let me ask you a question.
How many shells does promethium have?
It’s 6. Right?
You have already seen the bohr model of promethium atom in the above table.
From the Bohr model, it can be found that the number of orbits or shells in promethium is 6. Hence, as promethium has 6 orbits, it lies in period 6 of the Periodic table.
Why is Promethium in f-block?
Before knowing this reason, first of all I want to ask you a simple question.
How can you determine the blocks-wise position of elements?
The simple answer: The elements will lie in the s, p, d or f block will completely depend upon the subshell in which the last electron will enter.
For example; the electron configuration of promethium is [Xe] 6s2 4f5.
So the last electron of promethium enters the f-subshell or f-orbital.
Hence, promethium is the f-block element.
5 Interesting facts about Promethium
Interesting facts about promethium element are mentioned below.
- Promethium is mostly found as a byproduct of the uranium fission reactions.
- Promethium was discovered by 3 chemists Lawrence Glendenin, Jacob Marinsky, Charles Coryell (in 1945).
- The existence of promethium was predicted by a chemist, Bohuslav Brauner in 1902.
- The name “promethium” was suggested by Charles Coryell’s wife (Grace Mary Coryell).
- Out of all the rare earth metals, the promethium was the last discovered rare earth metal.
Properties of Promethium
The physical and chemical properties of promethium element are mentioned below.
Physical properties of Promethium
Physical properties of promethium are mentioned below.
- Promethium is silvery metallic in appearance and the salts of promethium glow with a greenish or bluish light in the dark.
- The melting point of promethium is 1042 °C and its boiling point is 3000 °C.
- The crystal structure of promethium is Double hexagonal close packed.
- The atomic mass of the most stable isotope of promethium is 145 u and its density is 7.26 g/cm3.
- Promethium has two naturally occurring isotopes, and they are 145Pm and 147Pm. Both of these isotopes are present in very trace amounts.
Chemical properties of Promethium
Chemical properties of promethium are mentioned below.
- The isotopes of promethium are radioactive and it undergoes beta-decay.
- The common oxidation state of promethium is +3, and this oxidation state is seen in most of the lanthanides.
- The electron configuration of promethium is [Xe] 6s2 4f5 which indicates that the last electron enters the f-orbital. Because of this reason, it is a f-block element.
- The first ionization energy of promethium is 5.55 eV.
Uses of Promethium
Uses of promethium are mentioned below.
- Promethium is a very rare element and hence it does not have more uses. It is generally used for research work.
- Promethium is used in nuclear batteries that are used in guided missiles as well as spacecraft.
- There are possibilities that it can be used in medical devices.
Explore our New Interactive Periodic Table (with Rotating Bohr Models and More)

Details about this Periodic table:
- Access detailed info on all elements: atomic mass, electron configurations, charges, and more.
- View rotating Bohr models for all 118 elements.
- Get a free HD image of the Periodic Table.
Note: For future use, bookmark this Periodic table or visit “PeriodicTableGuide.com”
External resources:
- Promethium – Element information, properties and uses | Periodic Table. (n.d.). Promethium – Element Information, Properties and Uses | Periodic Table. https://www.rsc.org/periodic-table/element/61/promethium
- Promethium – Wikipedia. (2012, June 20). Promethium – Wikipedia. https://en.wikipedia.org/wiki/Promethium
- P. (n.d.). Promethium | Pm (Element) – PubChem. Promethium | Pm (Element) – PubChem. https://pubchem.ncbi.nlm.nih.gov/element/Promethium
- It’s Elemental – The Element Promethium. (n.d.). It’s Elemental – the Element Promethium. https://education.jlab.org/itselemental/ele061.html
- University of Sheffield and WebElements Ltd, M. W. (n.d.). WebElements Periodic Table » Promethium » the essentials. WebElements Periodic Table &Raquo; Promethium &Raquo; the Essentials. https://www.webelements.com/promethium/
- Periodic Table of Elements: Los Alamos National Laboratory. (n.d.). Periodic Table of Elements: Los Alamos National Laboratory. https://periodic.lanl.gov/61.shtml
- Periodic Table of Nottingham – University of Nottingham. (n.d.). Periodic Table of Nottingham – University of Nottingham. https://www.nottingham.ac.uk/periodicnottingham/promethium
