This is a SUPER easy guide on Californium element.
In fact, the table mentioned below is the perfect information box (Which gives you every single detail about the Californium element in Periodic table.)
So if you want to know anything about Californium element, then this guide is for you.
Let’s finish this very quickly.
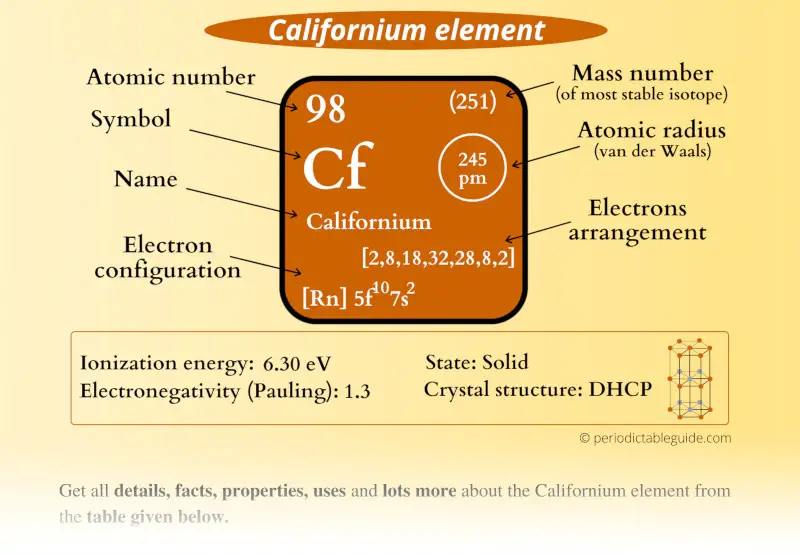
Californium Element (Cf) Information
| Appearance |  Silvery |
| State (at STP) | Solid |
| Position in Periodic table | 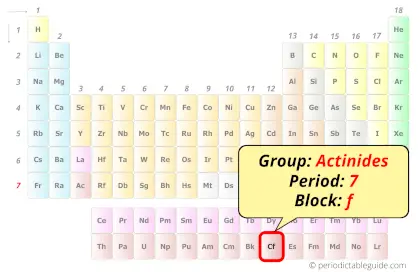 Group: actinides, Period: 7, Block: f |
| Category | 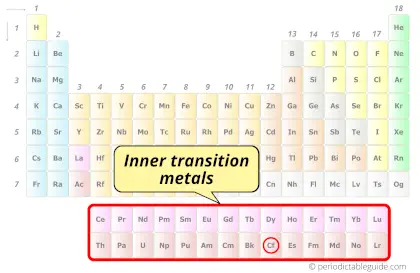 Inner transition metals |
| Atomic number or Protons | 98 |
| Neutrons | 153 |
| Electrons | 98 |
| Symbol | Cf |
| Atomic mass of Californium (most stable isotope) | 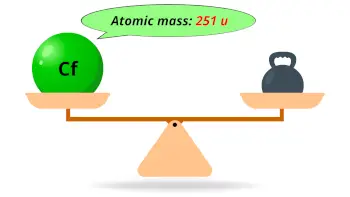 251 u |
| Electrons arrangement or Bohr model | 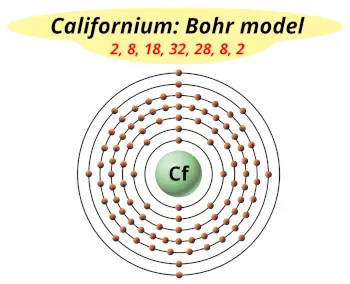 2, 8, 18, 32, 28, 8, 2 |
| Electronic configuration | [Rn] 5f10 7s2 |
| Atomic radius | 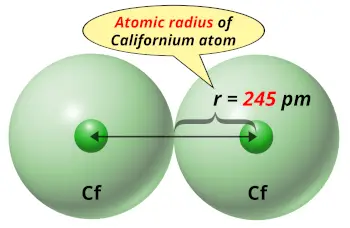 245 picometers (van der Waals radius) |
| 1st Ionization energy | 6.30 eV |
| Electronegativity | 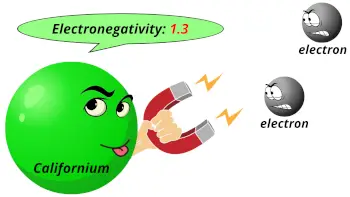 1.3 (Pauling scale) |
| Crystal structure | 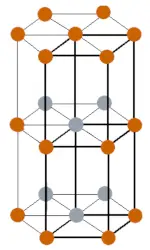 DHCP (Double hexagonal close packed) |
| Melting point | 1173 K or 900 °C or 1652 °F |
| Boiling point | 1743 K or 1470 °C or 2678 °F |
| Density | 15.1 g/cm3 |
| Who discovered Californium and when? | Glenn T. Seaborg, Stanley Thompson, Albert Ghiorso and Kenneth Street (in 1950) |
| CAS number | 7440-71-3 |
Californium in Periodic table
Californium element is in period 7 and in actinides group of the Periodic table. Californium is the f-block element and it belongs to inner transition metals group.
| H | He | ||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La* | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac** | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| *Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | ||||
| **Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
←Move to: Berkelium (Bk) element – Periodic Table
→Move to: Einsteinium (Es) element – Periodic Table
Why is Californium in Period 7?

Let me ask you a question.
How many shells does californium have?
It’s 7. Right?
You have already seen the bohr model of californium atom in the above table.
From the Bohr model, it can be found that the number of orbits or shells in californium is 7. Hence, as californium has 7 orbits, it lies in period 7 of the Periodic table.
Why is Californium in f-block?
Before knowing this reason, first of all I want to ask you a simple question.
How can you determine the blocks-wise position of elements?
The simple answer: The elements will lie in the s, p, d or f block will completely depend upon the subshell in which the last electron will enter.
For example; the electron configuration of californium is [Rn] 5f10 7s2.
So the last electron of californium enters the f-subshell or f-orbital.
Hence, californium is the f-block element.
5 Interesting facts about Californium
Interesting facts about californium element are mentioned below.
- The name californium came from the “California” from where it was discovered.
- Californium element was discovered by Glenn T. Seaborg, Stanley Thompson, Albert Ghiorso and Kenneth Street in 1950.
- There are 20 known isotopes of californium and all those isotopes are radioactive in nature.
- Out of all the radioactive isotopes of californium, the isotope 251Cf is the longest lived isotope and has a half-life of 898 years.
- Californium is a radioactive element that is prepared in nuclear reactors.
Properties of Californium
The physical and chemical properties of californium element are mentioned below.
Physical properties of Californium
Physical properties of californium are mentioned below.
- The pure californium metal is solid at room temperature and it is silvery wihte in appearance.
- Pure californium metal is a malleable and soft metal that can be cut with a razor blade.
- The estimated melting point and boiling points of californium are 900 °C and 1470 °C respectively.
- The californium element has DHCP (Double hexagonal close packed) crystal structure at lower temperatures, but it possess FCC (Face centered cubic) structure at higher temperatures.
Chemical properties of Californium
Chemical properties of californium are mentioned below.
- Californium is chemically reactive metal and it tarnishes slowly in the air.
- If the moisture content of the air is more, then the tarnishing of californium will be rapid.
- Californium also reacts with gasses like hydrogen, nitrogen and chalcogens as well as it also reacts with aqueous mineral acids.
Uses of Californium
The uses of californium are mentioned below.
- Californium is used in applications of neutron emitters. 1 gram of californium can produce 139 million neutrons per minute.
- The isotope 252Cf is used to detect the amount of oil in the oil wells.
- 252Cf is also used in treatment of cervical cancer as well as brain cancer.
- Apart from all these uses, the californium element is also used in research work in chemistry.
Explore our New Interactive Periodic Table (with Rotating Bohr Models and More)

Details about this Periodic table:
- Access detailed info on all elements: atomic mass, electron configurations, charges, and more.
- View rotating Bohr models for all 118 elements.
- Get a free HD image of the Periodic Table.
Note: For future use, bookmark this Periodic table or visit “PeriodicTableGuide.com”
External resources:
- P. (n.d.). Californium | Cf (Element) – PubChem. Californium | Cf (Element) – PubChem. https://pubchem.ncbi.nlm.nih.gov/element/Californium
- It’s Elemental – The Element Californium. (n.d.). It’s Elemental – the Element Californium. https://education.jlab.org/itselemental/ele098.html
- Californium – Wikipedia. (2011, July 11). Californium – Wikipedia. https://en.wikipedia.org/wiki/Californium
- Californium – Element information, properties and uses | Periodic Table. (n.d.). Californium – Element Information, Properties and Uses | Periodic Table. https://www.rsc.org/periodic-table/element/98/californium
- Californium | CCDC. (n.d.). Californium | CCDC. https://www.ccdc.cam.ac.uk/elements/californium/
- Thompson, S. G., Street, K., Ghiorso, A., & Seaborg, G. T. (1950, December 1). The New Element Californium (Atomic Number 98). Physical Review, 80(5), 790–796. https://doi.org/10.1103/physrev.80.790
