This is a SUPER easy guide on Tellurium element.
In fact, the table mentioned below is the perfect information box (Which gives you every single detail about the Tellurium element in Periodic table.)
So if you want to know anything about Tellurium element, then this guide is for you.
Let’s dive right into it!
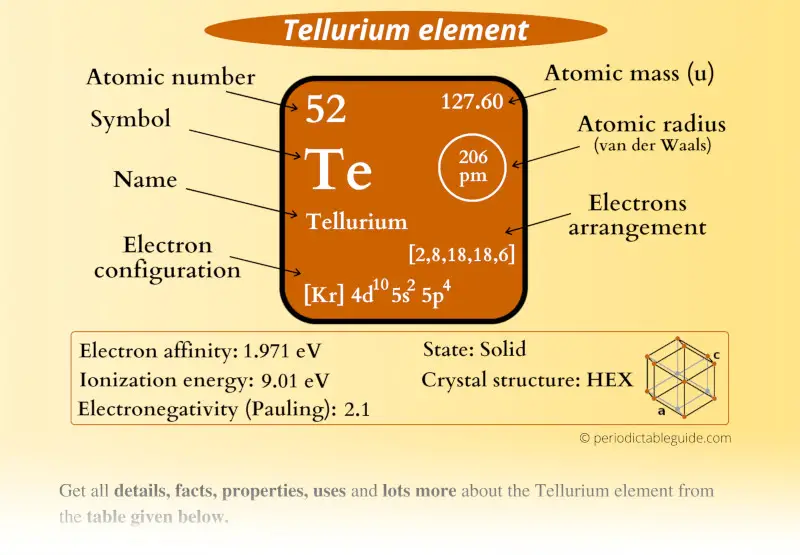
Tellurium Element (Te) Information
| Appearance |  Crystalline form is: Silvery gray lustrous Amorphous form is: Brown-black powder |
| State (at STP) | Solid |
| Position in Periodic table | 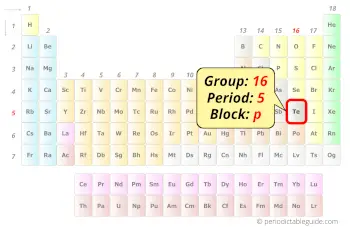 Group: 16, Period: 5, Block: p |
| Category |  Chalcogens |
| Atomic number or Protons | 52 |
| Neutrons | 76 |
| Electrons | 52 |
| Symbol | Te |
| Atomic mass | 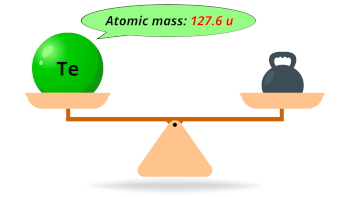 127.6 u |
| Electrons arrangement or Bohr model | 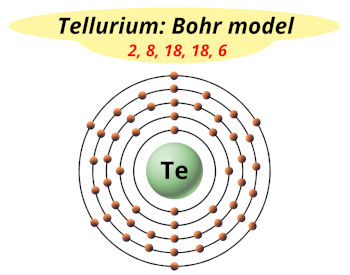 2, 8, 18, 18, 6 |
| Electronic configuration | [Kr] 4d10 5s2 5p4 |
| Atomic radius | 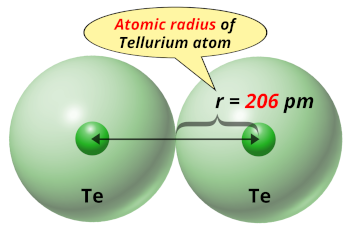 206 picometers (van der Waals radius) |
| Valence electrons | 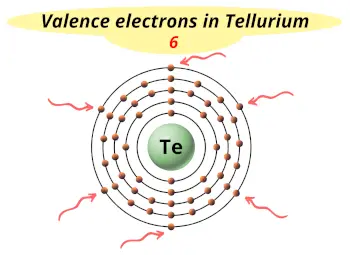 6 |
| 1st Ionization energy | 9.01 eV |
| Electronegativity | 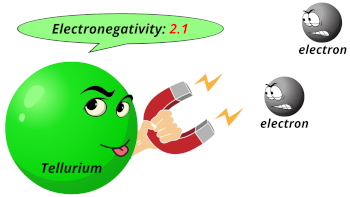 2.1 (Pauling scale) |
| Crystal structure | 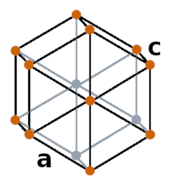 Hexagonal |
| Melting point | 722.6 K or 449.5 °C or 841.1 °F |
| Boiling point | 1261 K or 988 °C or 1810 °F |
| Density | 6.24 g/cm3 |
| Main isotope | 130Te |
| Who discovered Tellurium and when? |  Franz-Joseph Müller von Reichenstein (in 1782) |
| CAS number | 13494-80-9 |
Tellurium in Periodic table
Tellurium element is in group 16 and period 5 of the Periodic table. Tellurium is the p-block element and it belongs to chalcogens group.
| H | He | ||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La* | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac** | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| *Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | ||||
| **Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
←Move to: Antimony (Sb) element – Periodic Table
→Move to: Iodine (I) element – Periodic Table
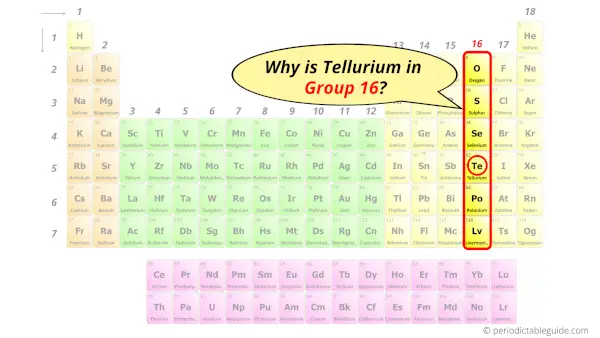
Why is Tellurium in Group 16?
Do you know, how many electrons can be accommodated in the first shell, second shell, third shell, fourth shell, etc…?
Here is the table showing the capacity of orbits to hold electrons.
Number of electrons in shells.
| Orbit / Shell (n) | Maximum no. of electrons this orbit can hold (2 × n2) |
| K shell, n = 1 | 2 × 1² = 2 |
| L shell, n = 2 | 2 × 2² = 8 |
| M shell, n = 3 | 2 × 3² = 18 |
| N shell, n = 4 | 2 × 4² = 32 |
| . . . | . . . |
Thus,
- 1st shell can hold 2 electrons.
- 2nd shell can hold 8 electrons.
- 3rd shell can hold 18 electrons.
- 4th shell can hold 32 electrons, and so on.
Now the atomic number of tellurium (Te) is 52.
Hence the electron arrangement in tellurium is 2, 8, 18, 18, 6. And the electron configuration of tellurium is 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s2 5p4.
This electron arrangement and electron configuration indicates that the outermost orbit (i.e orbit number 5) of tellurium atom has 6 electrons.
Hence, it lies in group 16.
Why is Tellurium in Period 5?

Let me ask you a question.
How many shells does tellurium have?
It’s 5. Right?
You have already seen the bohr model of tellurium atom in the above table.
From the Bohr model, it can be found that the number of orbits or shells in tellurium is 5. Hence, as tellurium has 5 orbits, it lies in period 5 of the Periodic table.
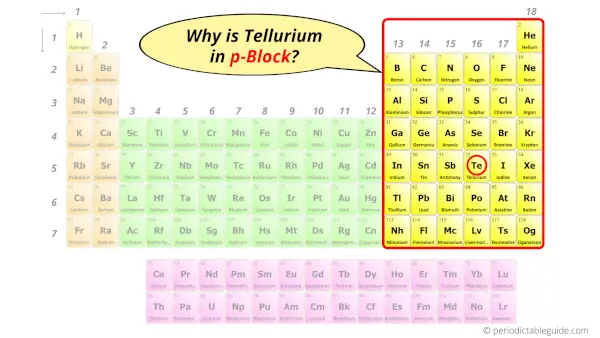
Why is Tellurium in p-block?
Before knowing this reason, first of all I want to ask you a simple question.
How can you determine the blocks-wise position of elements?
The simple answer: The elements will lie in the s, p, d or f block will completely depend upon the subshell in which the last electron will enter.
For example; the electron configuration of tellurium is [Kr] 4d10 5s2 5p4.
So the last electron of tellurium enters the p-subshell or p-orbital.
Hence, tellurium is the p-block element.
6 Interesting facts about Tellurium
Interesting facts about tellurium element are mentioned below.
- The name tellurium was derived from the latin word “tellus”, which means earth.
- Tellurium was discovered by Franz-Joseph Müller von Reichenstein in 1782.
- Tellurium is a metalloid that shows few properties of metal and few properties of nonmetals.
- Tellurium is rarely found in its natural form, but it is mostly found in a mineral form with gold and other metals.
- China is the leading producer of tellurium in the world.
- Tellurium is also obtained during the refining of copper. Approximately 1 pound of tellurium is obtained by refining around 550 tons of copper.
Properties of Tellurium
The physical and chemical properties of tellurium element are mentioned below.
Physical properties of Tellurium
Physical properties of tellurium are mentioned below.
- The pure form of tellurium has a silver white metallic lustre.
- The atomic mass of tellurium is 127.6 u and its density is 6.24 g/cm3.
- The melting point of tellurium is 449.5 °C and its boiling point is 988 °C.
- The crystal structure of tellurium is hexagonal.
- Tellurium has many isotopes, and out of them the most abundant isotope is 130Te (around 34%).
Chemical properties of Tellurium
Chemical properties of tellurium are mentioned below.
- When tellurium is heated in the presence of air, it burns with a greenish-blue flame.
- Tellurium reacts with oxygen at higher temperature and forms tellurium dioxide (TeO2).
- In the compound state, tellurium generally exists in the oxidation states +4 or +5.
- The 1st ionization energy of tellurium is 9.01 eV.
- Electronegativity is tellurium is 2.1 on Pauling scale.
Uses of Tellurium
Uses of tellurium are mentioned below.
- Tellurium is added to copper and steel to improve their machinability.
- Tellurium is also added to castings as well as it is also used in ceramics.
- When tellurium is added to lead, it increases its corrosion resistance property as well as it improves its strength.
- Tellurium is also used in making solar panels, which increases the efficiency of solar panels.
- Superconductors are also made by alloying tellurium with cadmium and mercury.
Explore our New Interactive Periodic Table (with Rotating Bohr Models and More)

Details about this Periodic table:
- Access detailed info on all elements: atomic mass, electron configurations, charges, and more.
- View rotating Bohr models for all 118 elements.
- Get a free HD image of the Periodic Table.
Note: For future use, bookmark this Periodic table or visit “PeriodicTableGuide.com”
External resources:
- Periodic Table of Elements: Los Alamos National Laboratory. (n.d.). Periodic Table of Elements: Los Alamos National Laboratory. https://periodic.lanl.gov/52.shtml
- Medina-Cruz, D., Tien-Street, W., Vernet-Crua, A., Zhang, B., Huang, X., Murali, A., Chen, J., Liu, Y., Garcia-Martin, J. M., Cholula-Díaz, J. L., & Webster, T. (2020). Tellurium, the Forgotten Element: A Review of the Properties, Processes, and Biomedical Applications of the Bulk and Nanoscale Metalloid. Racing for the Surface, 723–783. https://doi.org/10.1007/978-3-030-34471-9_26
- Tellurium – Wikipedia. (2010, January 16). Tellurium – Wikipedia. https://en.wikipedia.org/wiki/Tellurium
- Tellurium – Element information, properties and uses | Periodic Table. (n.d.). Tellurium – Element Information, Properties and Uses | Periodic Table. https://www.rsc.org/periodic-table/element/52/tellurium
- It’s Elemental – The Element Tellurium. (n.d.). It’s Elemental – the Element Tellurium. https://education.jlab.org/itselemental/ele052.html
- P. (n.d.). Tellurium | Te (Element) – PubChem. Tellurium | Te (Element) – PubChem. https://pubchem.ncbi.nlm.nih.gov/element/Tellurium
