This is a SUPER easy guide on Chromium element.
In fact, the table mentioned below is the perfect information box (Which gives you every single detail about the Chromium element in Periodic table.)
So if you want to know anything about Chromium element, then this guide is for you.
Let’s dive right into it!
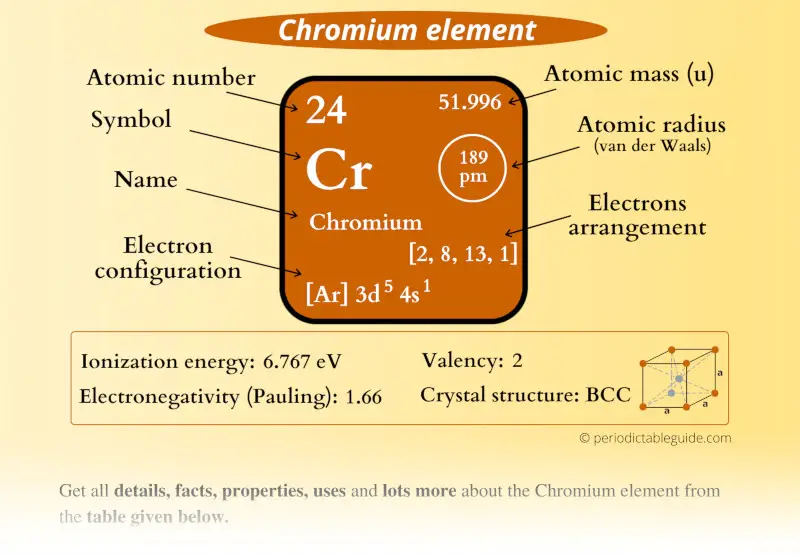
Chromium Element (Cr) Information
| Appearance |  Silvery metallic color |
| State (at STP) | Solid |
| Position in Periodic table | 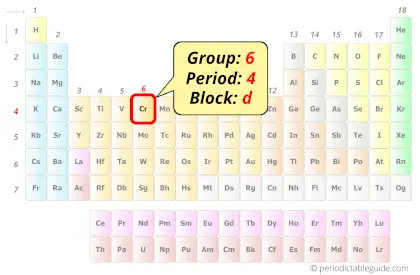 Group: 6, Period: 4, Block: d |
| Category | 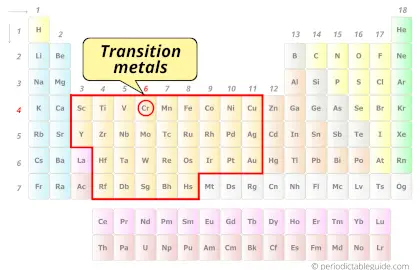 Transition metals |
| Atomic number or Protons | 24 |
| Neutrons | 28 |
| Electrons | 24 |
| Symbol | Cr |
| Atomic mass |  51.996 u |
| Electrons arrangement or Bohr model | 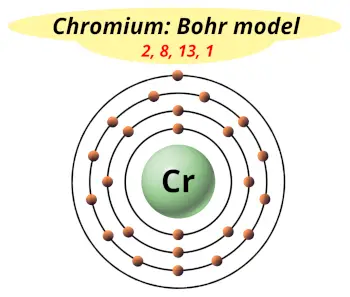 2, 8, 13, 1 |
| Electronic configuration | [Ar] 3d5 4s1 |
| Atomic radius | 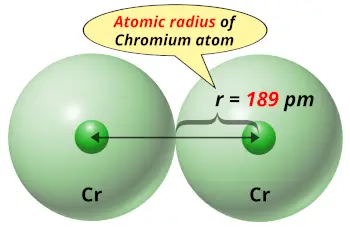 189 picometers (van der Waals radius) |
| 1st Ionization energy | 6.767 eV |
| Electronegativity | 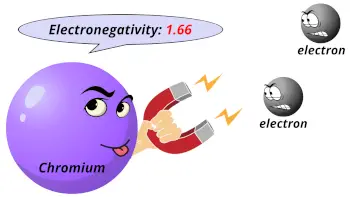 1.66 (Pauling scale) |
| Crystal structure | 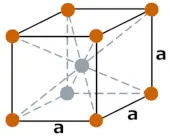 BCC (Body centered cubic) |
| Melting point | 2180 K or 1907 °C or 3465 °F |
| Boiling point | 2944 K or 2671 °C or 4840 °F |
| Density | 7.19 g/cm3 |
| Main isotope | 52Cr |
| Who discovered Chromium and when? |  Louis Nicolas Vauquelin in 1794 |
| CAS number | 7440-47-3 |
Chromium in Periodic table
Chromium element is in group 6 and period 4 of the Periodic table. Chromium is the d-block element and it belongs to transition metals group.
| H | He | ||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La* | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac** | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| *Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | ||||
| **Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
←Move to: Vanadium (V) element – Periodic Table
→Move to: Manganese (Mn) element – Periodic Table
Why is Chromium in Period 4?
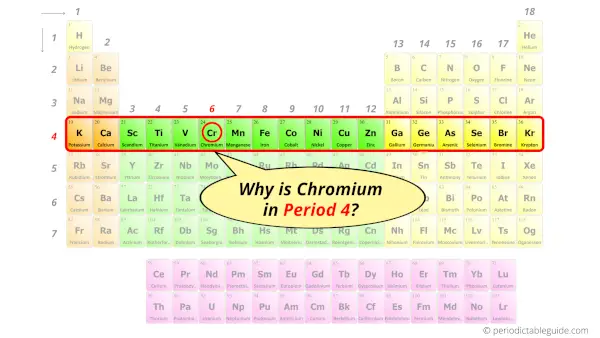
Let me ask you a question.
How many shells does chromium have?
It’s 4. Right?
You have already seen the bohr model of chromium atom in the above table.
From the Bohr model, it can be found that the number of orbits or shells in chromium is 4. Hence, as chromium has 4 orbits, it lies in period 4 of the Periodic table.
Why is Chromium in d-block?
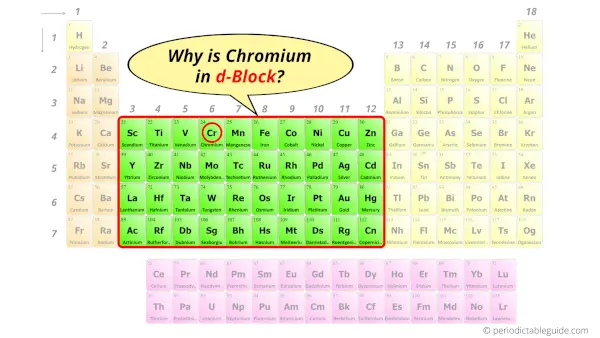
Before knowing this reason, first of all I want to ask you a simple question.
How can you determine the blocks-wise position of elements?
The simple answer: The elements will lie in the s, p, d or f block will completely depend upon the subshell in which the last electron will enter.
For example; the electron configuration of chromium is [Ar] 4s1 3d5.
So the last electron of chromium enters the d-subshell or d-orbital.
Hence, chromium is the d-block element.
Is Chromium a Transition Metal? Why?
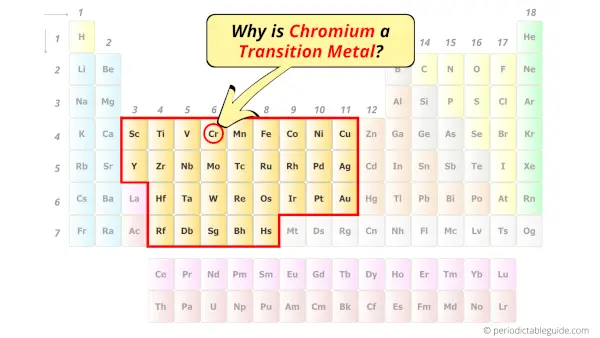
Yes, Chromium is a transition metal because it has incompletely filled d-orbital in its ground state.
Let me explain the exact meaning of this.
According to the definition of transition metals;
The element should compulsorily have incomplete d-orbitals, either in their ground state (M) or most common oxidation states (M1+, M2+, etc) then only they are called transition metals.
Now, the ground state of Chromium means its normal state in which it has neither gained nor lost any electron/s.
So the ground state of Chromium is Cr.
And the ground state electronic configuration of Chromium is [Ar] 4s1 3d5.
In this state, if we see the electron configuration of Chromium, then it possesses incomplete d-orbitals.
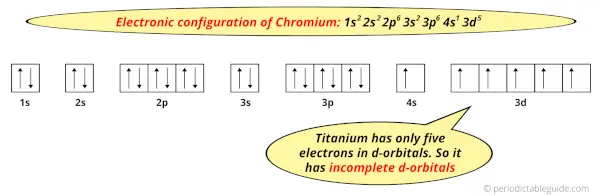
Because, there are only five electrons in the d-orbitals.
In order to have the complete d-orbitals, there must be 10 electrons in it.
But in the ground state electronic configuration of chromium, you can see that it has only 5 electrons in d-orbitals.
Thus, Chromium has incomplete d-orbitals.
And hence, as Chromium has incomplete d-orbitals, it is considered as a transition metal.
7 Interesting facts about Chromium
Interesting facts about chromium element are mentioned below.
- The name of chromium element is derived from the Greek word “chroma’ (meaning color).
- Chromium is found in the earth’s crust and it is the 21st most abundant element in the earth’s crust.
- The concentration of chromium in earth’s crust is around 100 ppm.
- The majority amount of chromium that is obtained is from the mineral chromite.
- South Africa is the leading producer of chromium in the world.
- The majority of chromium produced in the industries is used in making strong alloys.
- Chromium element is the 3rd hardest element found on the entire periodic table. (Carbon is the first and boron is the second).
Properties of Chromium
The physical and chemical properties of chromium element are mentioned below.
Physical properties of Chromium
Physical properties of chromium are mentioned below.
- Chromium is a strong metal having a silvery metallic lustre.
- Melting point of chromium is 1907 °C and its boiling point is 2671 °C.
- The atomic mass of chromium is 51.996 u and has a density of 7.19 g/cm3.
- There are many isotopes of chromium, out of which 52Cr is the most abundant (around 84%).
Chemical properties of Chromium
Chemical properties of chromium are mentioned below.
- Chromium is a fairly active metal and so it is not found in a free state. But it is always found as a compound with other elements.
- Chromium is a transition metal as it has incomplete d-orbitals.
- Chromium has anti corrosive properties due to which it offers a protective layer to the surface on which chromium is electroplated.
Uses of Chromium
Uses of chromium are mentioned below.
- Chromium is the most important metal that is used in stainless steel (which is used in manufacturing utensils). Stainless steels contain around 11% chromium.
- Chromium is used in electroplating of other metals. By electroplating, the thin layer of chromium is applied to other metals.
- Chromium plating is used in coating of car parts, home appliances, etc to give a shiny finish.
- The heat resistance bricks used in furnaces contain chromium in it, due to which the bricks retain strength at higher temperature.
- Chromium is used in preparing pigments. Also the red color of ruby is due to the presence of chromium in it.
- Chromium salts are also used in leather tanning.
Explore our New Interactive Periodic Table (with Rotating Bohr Models and More)

Details about this Periodic table:
- Access detailed info on all elements: atomic mass, electron configurations, charges, and more.
- View rotating Bohr models for all 118 elements.
- Get a free HD image of the Periodic Table.
Note: For future use, bookmark this Periodic table or visit “PeriodicTableGuide.com”
External resources:
- Chromium – Element information, properties and uses | Periodic Table. (n.d.). Chromium – Element Information, Properties and Uses | Periodic Table. https://www.rsc.org/periodic-table/element/24/chromium
- Chromium – Wikipedia. (2018, October 11). Chromium – Wikipedia. https://en.wikipedia.org/wiki/Chromium
- P. (n.d.). Chromium | Cr (Element) – PubChem. Chromium | Cr (Element) – PubChem. https://pubchem.ncbi.nlm.nih.gov/element/Chromium
- It’s Elemental – The Element Chromium. (n.d.). It’s Elemental – the Element Chromium. https://education.jlab.org/itselemental/ele024.html
- Chromium. (n.d.). Chromium. https://webbook.nist.gov/cgi/cbook.cgi?ID=7440-47-3&Units=SI
